Nutrition and Dietetic Product Shortages are A Neglected Issue in Alerting Systems and in Registries
Helena Jenzer, Sandra Büsser, Franziska Scheidegger- Balmer, Marta Silva and Leila Sadeghi
DOI10.4172/2472-1921.100022
1Health Division, Bern University of Applied Sciences, aR&D Nutrition and Dietetics, Murtenstrasse 10, 3008 Bern, Switzerland
2Faculty of Nutrition and Food Sciences of the University of Porto, Rua Dr. Roberto Frias, 4200-465 Porto, PortugalFaculty of Nutrition and Food Sciences of the University of Porto, Rua Dr. Roberto Frias, 4200-465 Porto, Portugal
- *Corresponding Author:
- Helena Jenzer
Health Division, Bern University of Applied Sciences
aR&D Nutrition and Dietetics
Murtenstrasse 103008 Bern, Switzerland
Tel: +41318484557
E-mail: helena.jenzer@bfh.ch
Received date: April 29, 2016; Accepted date: July 20, 2016; Published date: July 26, 2016
Citation: Jenzer H, Büsser S, Scheidegger-Balmer F, et al. Nutrition and Dietetic Product Shortages are A Neglected Issue in Alerting Systems and in Registries. J Clin Nutr Diet. 2016, 2:15. doi: 10.4172/2472-1921.100022
Abstract
Context: Medicines shortages have become a global phenomenon and are recognized to affect all health systems. It is a problem that has grown to become a crisis in terms of delivering patient care. Over 1400 products were reported to be in shortage in the Netherlands between 2004 and 2011. In Switzerland, in April 2016 as much as 221 products were on shortage. 44 alerts were noticed within the latest 7 days. Shortages induce individuals’ suffering, bad clinical outcomes and additional costs for public health.
Objective: The aim of this review is to evaluate whether nutrition and dietetic products are a neglected issue in shortages research.
Method: Eight medicines shortages reporting systems, ten batch recall registries, and homepages of manufacturers were evaluated. Categories such as food for special medical purposes and dietary use, supplements, and infant formula were included.
Results: Among the products in shortage, infant formula, micronutrient additives for parenteral nutrition, vitamin and trace elements products were most frequently affected. Infant formula and potassium chloride injection concentrates are repeatedly affected by shortages as well, mainly in Germany, since 2008. Only products for parenteral nutrition, vitamins, and trace elements for oral use are registered in medicines shortage registries.
Conclusion: It is highly desirable to include clinically used nutrition and dietetic products in registries. Artificial legal delimitation between food and medicines not reflecting natural commonalities impairs inclusion of nutritional products in reporting systems. It is likely that dietitians are not accus-tomed to report incidences to contribute to improved patient safety and to prevent further incidences of shortages. A rigid regulation revealed to be one of the driving forces for threat to shortage. The milder impact of the less rigid regulatory frame in food industry as compared to pharma industry is likely to facilitate an undisrupted supply chain.
Keywords
Nutrition; Dietetics; Foods for special medical purposes; Medicines; Shortages; Nutrients; Healthcare; Supply chain; Multidisciplinary collaboration; Shared responsibility; Action research; Systems research
Introduction
A global phenomenon
Medicines shortages (also referred to as drug shortages) are a challenging global phenomenon affecting all hospital and health systems [1-5]. Furthermore it is a phenomenon that if left alone threatens to become a crisis in terms of delivering patient care [6, 7].
There is no unanimously accepted shortage definition. Industry keeps on a tight understanding, i.e., in case of no availability even of alternatives. Carers however look at each registered and not available product as being on shortage. Not only, but mainly in oncology, specific products are needed for on-going multicycle therapies according to international protocols. Disruption and switch to an alternative is rarely feasible and com-promises seriously patient safety and clinical outcome [1, 8, 9].
Medicines shortages as a global phenomenon grew steadily and increased sharply in the USA within a few years from 2006 (70 shortages) to 2011 (267 shortages) [10]. In 2012, 99% of over 300 hospital pharmacists from 27 European countries had to cope with drug shortage problems. 63% of hospital pharmacists experienced it weekly, nowadays even daily, with an increasing frequency and magnitude of the problem. In the Netherlands first, followed by many other countries, they are monitored and published on a website. From 2004 to 2011, more than 1400 products were mentioned. The number increased from 91 in 2004 to 242 in 2011. In Belgium, in 2012 some 30 drugs were regularly unavailable [4, 11]. In Switzerland, the most recent information (May 2016) shows as much as 275 products on shortage with 44 alerts within the last 7 days [12]. Mean-while, in Europe not only isolated cases are in the focus, but examples representing all therapeutic groups. The average duration of a shortage increased from 139 to 242 days in the same period. Substitutions (62%), alternatives (25%) and compounding (2%) have been the methods of choice to cope with such situations [13]. To bridge a gap arising from a case of drug shortage will generally take from one to seven hours [14]. In any case, as a drug from the hospital formulary has been selected also for its favourable costbenefit ratio, alternatives are usually more costly compared to the standard product. A simple intermediate substitution of a drug on the formulary costs, on average, €1800, while a definite substitution somewhere between €3800 and €4690 (figures valid for Germany) [15].
Governments’ interventions
Already in 2011, the situation prompted authorities to intervene in the market and remind manufacturers and suppliers about their responsibility. US president Obama signed the Executive Order 13588 instructing the U.S. Food and Drug Administration (FDA) to require manufacturers to provide adequately advanced notices of discontinuation of certain prescription drugs as well as to review modifications of the production processes of these drugs more quickly. These requirements comprised of an obligation to notify and inform on drug shortages, but did not include a disclosure of the reasons nor of the decisions which led to a withdrawal of products from the market, except the cases where only one provider of a medically necessary active ingredient is available. These vital products had to be announced adequately if disruption was imminent. The FDA has created a task force for strategic planning and the European Medicines Agency (EMA) evaluates shortages caused by good manufacturing practice (GMP) compliance problems. As a result, in European countries 38 shortages were prevented in 2010, 195 in 2011, 282 in 2012 [16-20], 170 in 2013 and one year after 101 shortages could have been averted [21].
Causation
While most of the research, strategies and interventions to prevent shortages focus on medicines to treat cancer, infections, emergencies, cardio-vascular conditions, anesthetic products, and parenteral nutrition, improvements remained isolated and not globally successful, even though the causes of shortages are widely known [22]. The potential of causing shortages arises from a wide variety of factors, such as technical incidences, quality systems deficiencies, business management decisions, cultural and ethical values. Disruptions are known to occur on several steps and procedures along the supply chain (beginning at active pharmaceutical ingredients (API) and products manufacturing, and passing via economic and business decisions, to culminating in logistics including provision and procurement). There is a long history of quality issues such as heparin contamination, the propofol case, production line disruption in some plants after mergers, deregistrations due to uncovered registration cost, accidents such as disrupted cool transports in a blood donation transport case, demand spikes (the oseltamivir case) and many more. Parallel exports from low-price to high-price countries yield secondary local shortages in the exporting country [5].
As legislation seeks desperately to distinguish between food and pharmaceutical, a set of reflections has to be considered when evaluating the need of supervision of shortages. Nature itself does not distinguish between food and active pharmaceutical ingredient (API). Biochemical pathways are the same for both and their commonalities allow efficiently absorbing, distributing, metabolizing and excreting substrates, irrespective of their classification as either food or therapeutic agent.
Objective of the study
The aim of this article was to detect whether nutrition and dietetic products, such as food for special medical purposes, are sufficiently considered or rather a neglected issue.
Methods
To answer the question as to whether nutrition and dietetic products such as food for special medical purposes would be a neglected issue, as a first step a literature and data registry search for nutrition and dietetic products was conducted on national and international reporting systems of medicines shortages. Reference systems included [12, 18, 23-28]:
• European Medicines Agency (EMA)-Shortages Catalogue
• U.S. Food and Drug Administration (FDA)-The Drug Shortage Database
• American Society of Parenteral and Enteral Nutrition (A.S.P.E.N)- Register of Parenteral Nutrition Shortages
• The Swiss Drug Shortage Database
• The Canadian Drug Shortage Database
• American Society of Health-System Pharmacists (ASHP)
• Bundesinstitut für Arzneimittel und Medizinprodukt (BfArM)
• Institute of Hospital Pharmacy University Hospital Basel
The second step consisted in searches for case reports and product recalls in online food reporting systems of different European countries, Australia, New Zealand and the U.S. [29- 38]. Furthermore, a general web research on nutrients shortages and recalls mentioned in lists, registries, websites and patient comments on online blogs and helpdesks was performed between January and March 2016. In addition, current biomedical online databases such as PubMed, Cochrane, and Google Scholar were screened by two persons. MESH-terms and English and as well as German keywords comprised shortage, foods for special medical purposes; nutraceuticals, functional food, shortages, out of stock, shortage of enteral nutrition, rationing, parenteral nutrition, medicines shortages, enteral formula, parenteral formula, tube feeding, enteral feeding, oral nutritional supplements.
Foodstuff from agricultural production and food for healthy people was not considered, if no indication for therapeutic indication was obvious.
Results
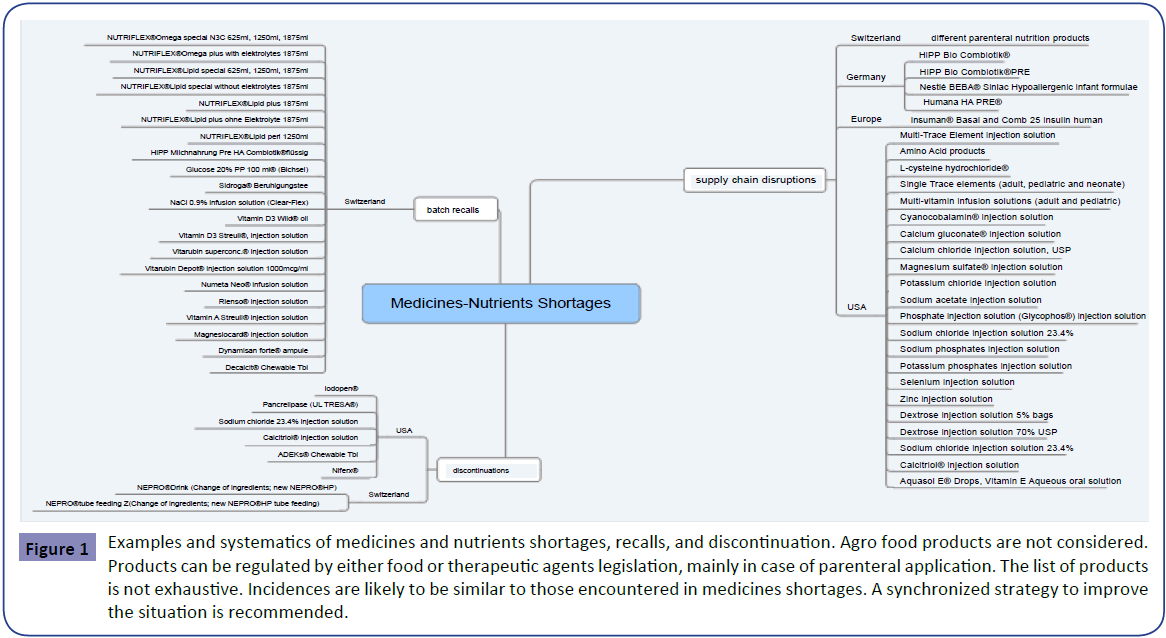
An overview of medicines and nutrients shortages retrieved from reference systems as disclosed under methods is depicted in Figure 1. This over-view is classified into shortages, recalls and discontinuations.
Figure 1: Examples and systematics of medicines and nutrients shortages, recalls, and discontinuation. Agro food products are not considered. Products can be regulated by either food or therapeutic agents legislation, mainly in case of parenteral application. The list of products is not exhaustive. Incidences are likely to be similar to those encountered in medicines shortages. A synchronized strategy to improve the situation is recommended.
Shortages of medicines for nutrition and dietetics purposes
Nutrition and dietetic products are known as special foods, which, in Switzerland, are regulated by a Federal Ordinance. This Ordinance defines admitted maximal concentrations of leading substances and further requirements. Beyond these limits, a product is considered as therapeutic agent. In comparison to ordinary foods of similar nature special foods differ in composition or method of manufacture. Special foods comprise foods for special medical purposes (FSMPs), low-content lactose or lactose-free foods, low protein foods, gluten-free foods, infant formula, foods for persons with increased energy and nutrient requirements, foods for weight management, and foods with added phytosterols or phytostanols and esters thereof [39]. Many products, mainly from phytochemical origin, are subject to delimitation agreements between food security and therapeutic agents authorities. The most important criteria to consider products either as drug or as food is given by its predominant use for pharmacotherapy instead of pure nutrition support for maintenance of body composition and function [40].
Special foods such as FSMPs are produced by food industry, not by pharma. Composition, labelling and sale of these products in Europe are regulated by the European Commission (Article 3 of Regulation (EU) No 609/2013) and controlled by national offices of each state. Among special foods, foods for special medical purposes (FSMPs) are used worldwide in all healthcare sections. They are intended for infants, children and adults with chronic medical conditions, malnutrition and for individuals suffering or recovering from serious illnesses in case their nutritional needs cannot be covered by normal oral diet. FSMPs can either complete or replace oral diet in order to cover nutritional needs [41]. According to the European legislation, FSMP are determined for patients with limited, impaired or disturbed capacity to take, digest, absorb, metabolize or excrete ordinary food or certain nutrients contained therein, or for further medically indicated requirements [42]. FSMP should only be used under the supervision of healthcare professionals [43]. They also comprise nutritionally complete oral or tube feed formula, nutritionally complete and adapted formula such as middle-chain triglycerides (MCT) containing complete formula used for nutrition support of malabsorption and nutritionally incomplete products including thickeners, carbohydrate modules, fat modules and nutritionally altered formula designed to treat specific conditions such as phenylala-nine-reduced products in phenylketonuria.
Preliminary troubleshooting and coping strategies for medicines shortages such as reporting systems or provider-user negotiations have been implemented in many countries. However, products regulated by food legislation are only exceptionally found in registries, in contrast to products registered by medicines agencies or administrations such as Swissmedic, EMA, FDA. Products regulated as therapeutic agents are clearly more readily notified in registries if they are in shortage. Merely Nepro® Drink and Nepro® enteral tube feeding were found as examples of FSMPs shortage as a result of manufacturing discontinuation [44].
Shortages were found most frequent among products for parenteral nutrition. Bags for Total Parenteral Nutrition (TPN) as well as further injectables are not defined as FSMPs due to their iv application and pharmacotherapeutic indication [40]. From medical and nutritional points of view, parenteral nutrition shortages are of everyone’s concern. A study in 2011 conducted by the American Hospital Association (AHA) found that 89 % of the hospitals have experienced parenteral nutrition shortages [45]. All parenteral nutrition product types have occasionally been in short supply since spring 2010 in the US. The American Society for Parenteral and Enteral Nutrition (ASPEN) website provides an actual list of the current par-enteral nutrition component shortages in the U.S. registered on the FDA website. Case reports (in April 2016) for iodopen (Sodium Iodide injection), multivitamin injection for adults and pediatrics, potassium chloride injection, sodium chloride 23.4% injection, and sodium acetate injection were reported to be currently in shortage [23, 24]. On the ASHP and Canadian websites, amino acid concentrates, calcitriol injection, calcium chloride injection, calcium gluconate injection, dextrose injection solution 70% USP, L-cysteine hydrochloride injection, and sodium phosphate injection were reported as being in shortage since 2010 [26].
The Shortage Subcommittee of the Clinical Practice Committee of the American Society for Parenteral and Enteral Nutrition (ASPEN) has edited recommendations for product shortages, which help clinicians manage parenteral nutrition therapy during a time of unavailability of a standard product [46]. Albarano (2014) suggested the implementation of dual/redundant parenteral nutrition systems which may help to cope with supply interruptions and avoid the use of unfamiliar products and additional labor costs [47]. In the United States, most often only one company is manufacturing parenteral nutrition components. Importation of European products has to be approved by the FDA. A shortage arising from such a lag time was noticed in trace elements for adults and pediatrics in 2013 [48].
Infant formula and follow-on formula intended for feeding notbreast- fed infants (aged less than 12 month) are a further special foods class. Pro-cessed cereal-based foods and baby foods for infants and young children meet the nutritional requirements of infants (aged less than 12 month) and young children (aged less than 3 years) in good health. They are intended for infants while being weaned, and for young children as a supplement to diet and/or for adaptation to ordinary food [42]. Infant formula were affected repeatedly by disruptions of the supply chain in Germany and some European countries in summer 2013, autumn 2014 and autumn 2015 in case of HiPP PRE BIO Combiotik® and HiPP BIO Combiotik® Anfangsmilch. This information was found on the enterprise’s homepage only. HiPP consoled clients by offering an online infant formula order and home delivery service with limited order quantities. Later Hipp BIO Combiotik® Milks were recalled (cf. Recalls) [49-53].
Similarly, Nestlé BEBA Sinlac® Hypoallergenic infant formulae and Humana HA PRE® fell in a shortage situation. Nestlé BEBA Sinlac® infant products cannot be delivered to the USA for legislative reasons. Nestlé BEBA Sinlac® Hypoallergenic infant formulae suffered as well from a sup-ply interruption due to increased demand from emerging expanding markets such as China and further Asian countries. The exploding demand in Asian countries was caused by a scandal of an infant milk powder from the Chinese manufacturer Sanlu. Their milk powder for infants was stretched with melamine causing intoxications in thousands of infants [54-56]. In Europe this particular shortage affected mainly infants who suffered from allergies. Their mothers had to spend extra time and money to find alternative products. The risk of intolerance or allergies increases every time when an unknown product must be used. No recommendations nor guidelines were/are available for substitution of products for each other in a period of an acute shortage. Such shortages and recalls were communicated on the manufacturers’ website and Facebook.
On the ASHP websites, vitamin E aqueous oral solution were detected as being in shortage in 2014 [48]. Shortages of functional foods were not found. Functional foods are conventional foods consumed as part of a diet plan which may along with nutrition to provide health benefits [57]. Functional Foods contain bioactive compounds e.g. polyphenols in tea, beta-glucan in oats and barley or probiotics in yogurt. They may as well be enriched with bioactive compounds. Functional Foods has proven health benefits that reduce the risk of specific chronic diseases or beneficially affect target functions beyond its basic nutritional functions when consumed on a regular basis and in normal quantities. Nutraceuticals are the bioactive compounds which are extracted from the foods and consumed in the form of pills, tablets etc. They are intended to improve health and to reduce the risk of diseases in addition to normal diet. Proper regulatory oversight for nutraceuticals and functional foods by the European Legislation is still missing. The regulation about production, labeling and promotion of functional foods and nutraceuticals lays in the responsibility of the EU Member States [57, 58].
Recalls
B.Braun medical AG recalled several products for parenteral nutrition in 2011, i.e., Nutriflex Omega special® with electrolytes, Nutriflex Omega plus® with electrolytes, Nutriflex Lipid special® with electrolytes. Further recalls concerned Dynamisan forte®, many nutritive additives for infu-sions, i.e., Vitamin D3 Streuli®, Vitarubin Depot, Vitamin A Streuli®, Rienso®, and oral supplements, i.e., Vitamin D3 Wild® oil and Decalcit®, all registered with Swissmedic [29].
A weight loss product in 2013 called Albuterex®, Albutarex Xtreme®, Albutarex Femme® and Atkins Advantage Chocolate Decadence Bar® were recalled in Australia and New Zealand in 2005 [30]. Another batch recall in 2011 concerned GNC Women's Ultra® Mega/Active/Energy and Me-tabolism, prenatal formula with iron and multivitamins [37]. The main reasons for these food recalls referred to quality, missing ingredient and allergen labelling as well as composition out of specification.
HiPP Pre HA Combiotik® fluid was recalled in 2014 due to quality problems [49-51]. Products affected by supply disruptions in Germany since 2008 were special infant foods HiPP PRE BIO Combiotik® and HiPP BIO Combiotik® Anfangsmilch and a recall of HiPP Pre HA Combiotik®fluid. According to the producers disruption of the supply chain was due to an increased demand from China. Batch recall of the HiPP Pre HA Combiotik®fluid was required as a result of a lower amount of Vitamin B12 than intended.
Nutraceuticals recalls were found occasionally on product recall portals for clients like the Veganz® Protein powder or the Bio Moringa® capsules and powder [33, 36].
Discontinuations
Unapproved replacement of pancreatic enzymes other than Pancrease MT® were not allowed to be marketed or distributed after April 28, 2010 when Ethex, Major, and X-Gen have discontinued manufacturing these unapproved products. Solvay discontinued Creon® 5, Creon® 10, Creon® 20 to promote the approved Creon® product [58]. Other products such as Ultresa® (Pancrelipase), Viokace®, Pertzye® were discontinued for a period of two years until they were approved by the FDA. Ultresa® (Pancreas lipase) was used by children and people with cystic fibrosis, amongst others. After discontinuation of the drug’s supply from 2010 until 2012, the patients had to switch to another product which received FDA approval. Upon switching products, many patients reported weight loss [59].
ADEKs Chewable Tablets and Niferex® (an iron polysaccharide oral liquid) were not available as a result of manufacturing discontinuation. Discontinuation of ADEKs Chewable Tablets and further multivitamin production stops caused a secondary shortage in water-miscible oral multiple vitamins for a certain period due to a spike demand [28].
Discussion
As many as 26 definitions for drug/medicines shortages are stipulated by legislations, governmental and professional organizations, scientific articles and national and international databases. A current, single and uniform definition of drug shortages is needed [60, 61].
Nutrition and dietetic products are rarely recorded in medicines shortages registries most likely because they are regulated less strictly by food law and therefore neglected. No shortage registries were found as related to foods for special medical purposes (FSMPs). However, food recall registers for consumers exist. As such products are frequently used by dietitian who are less frequently reporting incidence or shortages, such products tend to be included in shortages databases only if entered by the pharmacists’ channels. Although some products listed hereafter aren’t nutrients, they affect the nutritional state of patients and the flexibility of nutrition support teams. In order to warrant safe and professional care of the patients, being up to date in terms of nutrition and dietetic products is essential.
Registries of medicines shortages comprise parenteral nutrition (PN) bags and micronutrient supplements. One of the main reasons for PN short-ages could be the small return on investment which makes manufacturing PN bags unattractive and keeps stocks small (personal communications at LogiPharma 2016, Montreux, Switzerland). Therefore, only a limited number of PN manufacturers exist. They apply a just-in-time manufacturing model without relevant stocks. Therefore, PN are likely to fall frequently in shortage as a result of spike demands. In addition to small stocks, PN manufacturing is underlying to rigid GMP guidelines inducing similar causation for PN shortages as compared to the widely investigated drug shortages. These causation includes quality issues, active pharmaceutical ingredients (API) for injectables, loss of manufacturing sites due to natural disasters, product discontinuations, delays in production, and increased demand.
Shortages of FSMPs, such as enteral feeding and oral nutritional supplements (ONS) or functional foods have not been found. Shortages of enteral tube feeding products and ONS have neither been found on the manufacturer’s websites nor on batch recall lists. It either seems that these products are not affected by shortages as compared to PN bags, infant formula and oral vitamin and mineral supplements, or that enough immediately available similar alternatives from many producers including Abbott, Nestlé, Fresenius Kabi, Nutricia, Hipp, and Curatis exist. The question as to why more food producing enterprises put on the market a broad assortment of similar brands, as compared to non-blockbusters from pharma industry, might be related as well to the less rigid legislation and regulation imposed to food industry than that imposed to pharma industry. This facilitates commercial availability of food products.
FSMPs such as essential amino acid substituents for patients with phenylketonuria (PKU) are not communicated to be in shortage neither. They are produced as well by different manufacturers including Milupa Metabolics, Abbott, Vitaflo Pharma, Mead Johnson’s Products, MetaX, ComidaMed, Dr. Schär Medical Nutrition, Medotica Sagl, Thanaka, Nutrimedis, Nutricia, Groupe Nutrisens. The bigger number of small but independent production sites all over the world as compared to pharma industries might be an explanation for why there are few shortages even of orphan medicines and nutraceuticals for rare metabolic diseases.
Finally, shortage cases of PN and infant formula, oral nutritional supplements, as well as batch recalls of products for weight loss, protein powder and prenatal multivitamin formula underline the need for action. Although ASPEN has developed recommendations on how to troubleshoot PN shortages, there are still many nutrients in shortage for which no obvious solution exists yet. Guidelines on how to cope with drug shortages were edited also by the Swiss Association of Public Health Administration and Hospital Pharmacists (GSASA) [62]. In addition, supported by the most important stakeholders such as Swiss Associations and Federations of pharmacists (Swisspharma), physicians (FMH), and hospitals (H+), an agreement with the leading associations of pharmaceutical industry (ASSGP, Intergenerica, Interpharma, scienceindustries, and vips) was adopted in order to readily make available active ingredients for individualized preparations and small scale stock production of commercially not available formulations or dosages [63]. Obviously, this has not been able to provide sustainable relief to the players at the end of the supply chain, i.e., the staff treating patients, the patients themselves, and the taxpayer having to finance an insufficiently effective system. The reason might be the progressive disappearance of hospital production in the last two decades. As a result, even if raw material could be procured, a manufacturing site disposing an authorization from inspectorates and supervising governmental institutions to produce medicines in small quantities for the own institution’s patients was not always ready to execute its responsibility [64].
Meanwhile, as of 2016, the situation, mainly for medicines shortages, is at its worst and favors uncoordinated initiatives [12]. Presumable linear and stable consumption should permit manufacturing and provision in time. From the users’ point of view, there is no convincing reason why such products should run out of stock. The recognized fact is that industry and wholesalers tend to keep capital bound in their stock as small as possible (personal oral communications at LogiPharma 2016, Montreux, Switzerland) most likely inspired by the electronic industry, which uses this strategy with an acceptable justification, i.e., the threat of new developments dramatically reducing monetary values of technically overridden products. However, medicines as well as nutritional and dietetics products are not threatened by equally short half-lives, thus the capital bound in a stock would not be lost as fast as electronic devices. In addition, medicines and nutritional/dietetics products have an actual direct impact on individuals’ health and quality of life and should for ethical reasons be available without disruption.
Whatever the problem for a true shortage might be, an adaptation and synchronization of the various points of view of the stakeholders along the supply chain is highly recommended. This is the reason why the proposal for the New COST Action CA15105 has been launched by independent researchers. It aims to create a multi-disciplinary partnership, platform and network of researchers from science, technology and business management in collaboration with the professionals from the relevant practice settings (represented by professional associations and manufacturing industry). Furthermore the project aims to facilitate exchange of coping strategies. It will also apply simulation approaches of decision-making in a multi-disciplinary complex context. This research approach of simulation of decision-making in complex systems will be beneficial to untangle inter-related activities in a complex supply system.
Strengths and limitations
This review focuses on nutrients and foodstuff relevant for clinical nutrition, e.g. parenteral nutrition, injectables, food for special medical purposes and dietary use, supplements, and infant formula. Agrofood products were not in the scope of this study. The commonality of these categories is their regulation by Good Manufacturing Practices (GMP) Guidelines as well as the limited number of production plants worldwide. Numerous ex-amples of quality issues occurring in food industries have been encountered, e.g. microbiological instability cases. However, such cases were not considered as not being relevant for clinical nutrition and as multiple alternatives being readily available for consumers. In addition, further medicines categories such as medical devices were not included in this review due to its intentional limitation to products for clinical nutrition. As classification of applicable laws is often a matter of court ruling, reflexions concentrated on the use rather than legislation and regulation.
Conclusion
Some products for nutrition support and therapy are seriously affected by shortages. However, artificial legal delimitation between food and medicines not reflecting natural commonalities impairs inclusion of nutritional products in reporting systems. It is likely that dietitians are not yet entirely used to report incidences in a way, which is established in medical professions such as physicians and pharmacists. Further, it is likely that more groups of medicines are a neglected issue as well, such as medical devices.
Round table negotiations gathering all key players and stakeholders are the main option to improve the supply chain disruptions in times of serious economic and regulatory restrictions for all the players on the healthcare scene. In addition, the impact of the more rigid regulatory frame for the pharma industry as compared to the food industry has to be closely evaluated as a main driver of shortages, as pressure provokes escape behaviours. The milder impact of the less rigid regulatory frame in food industry as compared to pharma industry is likely to facilitate an undisrupted supply chain.
Acknowledgement
This article is based upon work from COST Action CA 15105 European Medicines Shortages Research Network-addressing supply problems to patients (Medicines Shortages), supported by COST (European Cooperation in Science and Technology).
References
- McLaughlin M (2013) Effects on Patient Care Caused by Drug Shortages. JMCP 19:783-788.
- Lynas K (2013) Patient care and health compromised by drug shortages in Canada. Can Pharmacists J 146:67-68.
- (2012) FIP calls attention to medicines shortages.
- EAHP (2013) 99% of HPs experience medicines shortages in past year.
- Jenzer H, Fenton MV (2015) Availability of medicines. In: Bouwman-Boer Y, Fenton-May V, LeBrun P (eds.) Practical Pharmaceutics. An International Guideline for the Preparation, Care and Use of Medicinal Products. Springer International,pp:25-50.
- McLaughlin M (2013) Empty Shelves, Full of Frustration. Hosp Pharmacy 48: 617-618.
- The Pharmaceutical Journal (2014) Rationing of chemotherapy in Europe a possibility if shortages problem not resolved, hospital pharmacist congress hears. Pharmac J 292/7804: 357.
- Center for Drug Evaluation and Research (2012) Manual of policies and procedures. MAPP.
- Kaakeh R (2011) lmpact of drug shortages on U.S. health systems. Am J Health System Pharm 68:1811-1819.
- Meyer T (2012) The Anatomy of the Drug Shortages. APSF Newsletter 27:7-9.
- Huys I, Simoens S (2013) European drug shortages: a call for action! Int J Pharmacy Practice 21:1-2.
- Martinelli Consulting (2016) The database of supply shortages of drugs in Switzerland.
- Postma D, de Wolf L (2013) Royal Dutch Pharmacists Association KNMP. Drug shortages in The Netherlands: monitored by Farmanco.18th Congress of the EAHP, 13–15 Mar 2013, Paris.Poster DSL 008.Eur JHosp Pharm SciPract 20: A90.
- Deutschmann W (2005) Supply shortages, production settings and imports (Part 1). Krankenhauspharmazie 26:14-19.
- Fischer BR (2013) Drug changes: Save not always worthwhile.DtschÄrzteblatt 110:A-120/B-206/C-206
- The White House (2011) Executive Order 13588–Reducing Prescription Drug Shortages.
- (2014) Food and Drug Administration Safety and Innovation Act (FDASIA).
- European Medicines Agency (2016) Shortages catalogue.
- Klein JO, Myers MG (2006) Vaccine Shortages: Why They Occur and What Needs to Be Done to Strengthen Vaccine Supply. Pediatrics 117: 2269-2275.
- Frontini R (2014) Speech at the launch of the 2014 survey on medicines shortages in Europe.
- (2016) U.S. Food and Drug Administration.
- Birgli® Report (2014) ISPE Drug Shortages Prevention Plan.
- U.S. Food and Drug Administration (2016) Drug shortages Database.
- American Society of Parenteral and Enteral Nutrition (2016) Product Shortages.
- (2016) The Canadian Drug Shortage Database.
- American Society of Health-System Pharmacists (2016) Drug Shortages.
- Federal Institute for Drugs and Medical products (2016) Delivery bottlenecks for human medicines in Germany.
- Hospital-Pharmacy University Hospital Basel (2016) Overview of current delays/below breaks and call backs.
- Swissmedic batch (2016)Recall lists 2006-2016.
- (2016) Food Standards Australia, New Zealand.
- (2016) U.S. Department of Health & Human Services.
- United States Department of Agriculture (2016) Food Safety and Inspection Service.
- (2016)Recall Portal for Germany.
- AGES (2016)Produktwarnungen&Produktrückrufe.
- European Commission (2016) Rapid Alert System for Food and Feed (RASFF).
- The Portal of the federal states and the BVL (2016) Food Alerts.
- Australian Competition and Consumer Commission (2016) Product Safety Recalls Australia.
- (2016) Global portal on product recalls.
- Federal Department of Home Affairs (2005) Ordinance on special food from 23, Swiss.
- Swissmedic (2016) Selected boundary issues.
- European Food Safety Authority (2016) FAQ: Foods for special medical purposes.
- Regulation (EU) No 609/2013 on food intended for infants and young children, food for special medical purposes, and total diet replacement for weight control.
- Specialized Nutrition Europe (2016) Food for Special Medical Purposes.
- GHD Nutrition (2016) GmbH.
- American Hospital Association (2011) Survey on Drug Shortages.
- Mirtallo JM, Holcombe B, Kochevar M, Guenter P (2012) Parenteral nutrition product shortages: the A.S.P.E.N. strategy. NutrClinPract 27: 385-391.
- Albarano T (2014) Consideration for the use of a dual parenteral nutrition system.
- Corrigan ML (2013) Parenteral Nutrition Drug Shortages—AnOngoing but Solvable Problem. Today’s Dietitian 15: 46.
- HiPP (2015) Supply shortages.
- HiPP (2015) Delays HiPP Pre Organic Combiotik.
- HiPP (2014) Important information: recall of milk formula HA Pre Combiotik® liquid.
- HiPP(2015) Commercial availability.
- www.urbia.de/archiv/forum/th-4508412/hipp-bio-combiotik-pre-hamstern.html
- www.netmoms.de/fragen/detail/hipp-combiotik-dauernd-ausverkauft-umstieg-auf-hipp-bio-pre-23005440
- https://www.urbia.de/archiv/forum/th-1834216/wo-gibts-noch-sinlac-in-stl.html
- https://www.docsimon.co.uk/article/nestle-sinlac-650-g
- El Sohaimy SA (2012) Functional foods and nutraceuticalsmodern approach to food science. World Applied Sciences Journal 20: 691-708.
- ASHP (2012) Pancreatic Enzyme Replacement: Products containing Lipase, Protease, and Amylase.
- (2016) CysticFibrosis.com.
- Doyon M, Labrecque J (2008) Functional foods: a conceptual definition. Brit Food J 110: 1133-1149.
- De Weerdt E, Simoens S, Casteels M, Huys I (2015) Toward a European definition for a drug shortage: a qualitative study. Front Pharmacol 6: 253.
- GSASA (2016)Recommendations for dealing with supply shortages.
- GSASA (2011) Intent of the associations of the pharmaceutical industry in Switzerland against FMH, GSASA, H+ ,pharmaSuisse. GSASA e-News May 2013. Shortages of cancer drugs in the USA. Lancet Oncology 12: 313.
- Jenzer H (2012) A contribution to reanimate the perishing hospital pharmacy preparation. Eur J Hosp Pharm 19:477-478.
Open Access Journals
- Aquaculture & Veterinary Science
- Chemistry & Chemical Sciences
- Clinical Sciences
- Engineering
- General Science
- Genetics & Molecular Biology
- Health Care & Nursing
- Immunology & Microbiology
- Materials Science
- Mathematics & Physics
- Medical Sciences
- Neurology & Psychiatry
- Oncology & Cancer Science
- Pharmaceutical Sciences