Effect of a Polyherbal Dietary Supplement on Semen Characteristics: A Randomized, Double-Blind Placebo-Controlled Clinical Trial
Doug MacKay1*, Sonika Verma2 and Shalini Srivastava3
1 Department of Health Sciences, Leading Edge Health, McKenzie Avenue, Victoria, BC, Canada 2 Department of Science and communications, Vedic Life sciences Pvt. Ltd, Ace Business Center, Thane West, India 3 Department of Clinical Development, Vedic Lifesciences Pvt. Ltd, Ace Business Center, Thane West, Mumbai, Maharashtra, India
Published Date: 2022-05-09DOI10.36648/2472-1921.8.4.178
Doug MacKay1*, Sonika Verma2 and Shalini Srivastava3
1Department of Health Sciences, Leading Edge Health, McKenzie Avenue, Victoria, BC, Canada
2Department of Science and communications, Vedic Life sciences Pvt. Ltd, Ace Business Center, Thane West, India
3Department of Clinical Development, Vedic Lifesciences Pvt. Ltd, Ace Business Center, Thane West, Mumbai, Maharashtra, India
- *Corresponding Author:
- Doug MacKay
Department of Health Sciences, Leading Edge Health, McKenzie Avenue, Victoria, Canada
E-mail:doug@dmcontact.com
Received date: April 07, 2022, Manuscript No. IPJCND-22-13053; Editor Assigned date: April 14, 2022, PreQC No. IPJCND-22-13053(PQ);
Reviewed date: April 21, 2022, QC No. IPJCND-22-13053; Revised date: April 28, 2022, Manuscript No. IPJCND-22-13053(R); Published date: May 09, 2022, DOI: 10.36648/2472-1921.8.4.178
Citation: MacKay D, Verma S and Srivastava S (2022) Effect of a Polyherbal Dietary Supplement on Semen Characteristics: A Randomized, Double-Blind Placebo-Controlled Clinical Trial. J Clin Nutr Diet Vol.8 No.4: 178
Abstract
Context: Ejaculate volume, an important semen characteristic, plays a significant role in male fertility. There is a growing need to find supplements to increase ejaculate volume.
Objective: The primary objective of the study was to evaluate the efficacy and safety of a dietary supplement (SemenaxTM) on semen characteristics of hypospermic and normospermic men.
Design: A double-blind, randomized placebo-controlled exploratory clinical trial.
Setting: Six clinical sites 3 at Nasik, 2 at Pune and 1 at Mumbai, Maharashtra, India.
Participants: Males aged 30 Years-60 years (n=78) diagnosed with hypospermia/normospermia with low ejaculate volume; normozoospermia; mild to moderate oligozoospermia; impairment of sperm motility and morphology and erectile dysfunction.
Intervention: The participants were randomized in a ratio of 1:1 to receive the supplement (3200 mg in two divided doses) or placebo for two months.
Main outcome measures: Main outcomes were mean change in ejaculate volume, sperm characteristics, sexual function, and orgasm quality from baseline to end of the study compared to placebo. In addition the safety of the supplement was evaluated by questionnaires, vitals and laboratory parameters.
Results: The supplement significantly increased the ejaculate volume from the baseline values compared to the placebo (19.67% vs. 8%). The number of responders in the supplement group was significantly more than in the placebo group (50% vs. 16%). Orgasm quality was improved in 65.63% of participants compared to 41.9% in placebo. No major safety concerns were reported in both groups.
Conclusion: Overall, the dietary supplement effectively improved the ejaculate volume to significant levels without any significant adverse effects.
Keywords: Semen; Dietary supplement; Ejaculate volume; Hypospermia; Normospermia; Orgasm; Sexual function; Erectile dysfunction
Abbreviations: AE: Adverse Event; ANOVA: Analysis of Variance; CBC: Complete Blood Count; CONSORT; Consolidated Standards of Reporting Trials; ECG: Electrocardiogram; EDTA: Ethylenediamine Tetra-Acetic Acid; EF: Erectile Function; ESR: Erythrocyte Sedimentation Rate; ICH-GCP: International Conference on Harmonization-Good Clinical Practices; ICMR: Indian Council of Medical Research; IIEF: International Index of Erectile Function; IP: Investigational Products; ITT: Intention-To-Treat; PP: Per-Protocol; SD: Standard Deviation; SGPT: Serum Glutamic Pyruvic Transaminase; WHO: World Health Organization
Introduction
Ejaculate comprises the concentrated suspension of spermatozoa with L-carnitine and neutral alpha-glucosidase, stored in the paired epididymides (10% of the volume), diluted by prostatic secretions (10% of the final semen volume) containing acid phosphatase, citric acid, inositol, calcium, zinc and magnesium in the urethra and the fructose, ascorbic acid, and prostaglandins-containing seminal vesicle fluid (75%-80% of the final seminal fluid). A mucus-rich secrete from Cowpers' or bulbourethral glands forms a minor part of the ejaculate and is the first to appear during ejaculation [1-3]. Adequate ejaculate volume is necessary to transport sperms in the female reproductive tract for fertilization [4]. Also, the spermatozoa ejaculated in prostatic fluid preserve better motility and vitality than when expelled in seminal vesicular fluid. However, during the etiological screening of male infertility, the role of ejaculate volume as a causative factor is often neglected.
As per the World Health Organization (WHO), the ejaculate has two major quantifiable parameters for assessing any abnormality-the total number of spermatozoa per ejaculate and the fluid volume contributed by the various accessory glands. Nonetheless, the vitality, motility, and morphology of spermatozoa and the ejaculate composition are also essential for infertility investigation, but the stage of ejaculation and the type of environment (in vivo vs. in vitro) influences these two factors [2,4]. Other individual characteristics that can affect the ejaculate volume are the size of the testicles, endocrine status, intake of certain medications-prescribed (e.g., anti-hypertensives and anti-depressants), or over-the-counter supplements (e.g., anabolic steroids). The factors that can decrease the ejaculate volume can be classified as recommended by Roberts et al. i) Artifactual-short abstinence period and incomplete collection; ii) Psychological-anorgasmia; iii) Pathological-retrograde ejaculation-structural (damage to bladder neck) or functional (nerve and neurotransmitter), failure of emission (nerve), ejaculatory duct obstruction-congenital (± seminal vesicles or vas anomalies) or acquired, agenesis or aplasia of the seminal vesicles and prostate, seminal vesicle disease (infection, cysts, polycystic kidney disease), and hypogonadism [4].
Apart from improving fertility, several men perceive that increasing ejaculate volume increases orgasmic function and enhances sexual gratification though the exact mechanism behind this notion has not been clearly understood [5]. The management of low ejaculate volume mainly depends on its etiology. The role of pharmacotherapy is limited to functional abnormalities, while surgical therapy can alleviate structural abnormalities responsible for low ejaculatory volume. Thus there is a growing need to find alternatives to increase ejaculate volume. Supplements like lycopene, zinc, fish oil supplement, L-arginine, L-carnitine and specific polyherbal formulations are considered effective in increasing ejaculate volume [6-10]. Most of these studies have shown benefits on sperm concentration and characteristics (vitality, motility, and morphology), while only a few studies are available that concluded significant effects in increasing the ejaculate volume. However, many dietary supplements are available nowadays, which claim to increase ejaculate volume with their regular consumption. There is a dearth of robust scientific evidence to back up this claim or the effect of these products on orgasmic function. Therefore, there is an unmet medical need to conduct well-designed randomized controlled trials to substantiate such claims scientifically.
A polyherbal dietary supplement containing various herbs, vitamin E, amino acids and zinc was developed by Leading Edge Health, Canada to address the growing need for a safe and efficacious product to increase ejaculate volume. Unfortunately, perceived hypospermia has rarely been investigated, even in individuals with sexual problems. The present double-blind, randomized placebo-controlled study is an exploratory investigation to evaluate the efficacy and safety of the dietary supplement in men desiring to improve their semen quality. Furthermore, low sperm count is often associated with sexual dysfunctions like erectile dysfunction, reduced orgasmic quality and infertility. Therefore, the investigational product was also studied for its effect on sexual functioning and sperm characteristics in men with perceived hypospermia.
Methods
Study design
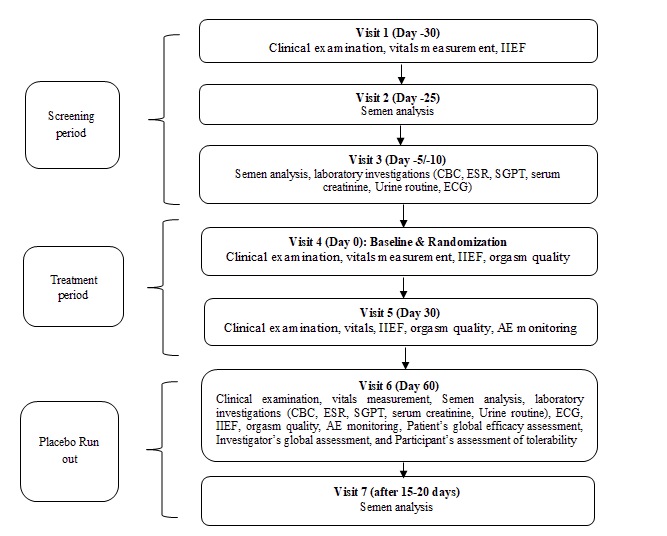
The present study was a randomized, double-blind, placebo-controlled, parallel-group, multi-center study to assess the efficacy and safety of the dietary supplement capsules on semen characteristics of hypospermic and normospermic men (n=78) after two months of oral administration. The main objectives of the study were to evaluate the efficacy of the dietary supplement on 1) The ejaculate volume of hypospermic and normospermic men, 2) Sperm characteristics including count, morphology, and motility, 3) Sexual function using the International Index of Erectile Function (IIEF), and 4) Orgasm quality. The secondary objective of the study was to assess the safety and tolerability of the dietary supplement in comparison to the placebo. For subgroup analysis the two groups were further stratified as hypospermic and normospermic. The details of the study flow and the visit schedule are provided in Figure 1 (Figure 1).
The study was conducted under the supervision of qualified physicians. The participants were recruited from the outpatient department of the selected six clinical sites: 3 at Nasik, 2 at Pune, and 1 at Mumbai, Maharashtra, India. After screening, the eligible participants were assigned to the supplement and control groups in a ratio of 1:1, using stratified block randomization. Randomly permuted blocks of 4 patients were generated using the statistical software Stats Direct Version 2.7.8 separately for each stratum (normospermic or hypospermic) by an independent researcher. The randomization codes were secured in tamper-evident sealed envelopes at the respective sites. Study participants and researchers remained blinded to the group assignments. In addition, an independent researcher not involved in the execution and analysis of the study undertook blinding procedures during manufacturing to ensure that the placebo and the supplement capsules were indistinguishable. Placebo and supplement capsules were matched for appearance and packed in identical containers with identical labels.
Participants
Participants who fulfilled all eligibility criteria listed in Table 1a and 1b were enrolled in the study (Tables 1a and 1b).
| Men aged 30 years-60 years, involved in a stable monogamous heterosexual relationship. |
| Hypospermia (semen volume lower than 2 ml) or normospermia (semen volume 2-5.5 ml) perceiving a reduction in the ejaculate. |
| Normozoospermia (sperm concentration>20 × 106 /ml) or mild to moderate oligozoospermia (sperm concentration 4-19.99 x 106/ml). |
| Mild to moderate impairment of sperm motility and sperm morphology. |
| Erectile dysfunction (IIEF-Erectile Function (IIEF-EF) score<26). |
| Having a response score of 3 and above to the IIEF-Orgasmic function (IIEF-OF) question. |
| Willing to maintain a constant sexual abstinence period of 2 days to 3 days before producing semen samples and comply with other semen collection procedures. |
Table 1a: Inclusion criteria.
| Aspermia (no semen) |
| Absence of fructose /low fructose (13 μmol per ejaculate). |
| Semen pH<7.2 or >8.0 |
| Excessive red blood cells (hemospermia). |
| Excessive leukocytes or leukospermia. |
| Severe impairment of ejaculate volume or sperm concentration or sperm motility or sperm morphology. |
| Ejaculate volume>5.5 ml (includes hyperspermia, i.e.,>7 ml of ejaculate volume). |
| Body mass index (BMI) ≥ 35 kg/m2 |
| Surgical history of radical prostatectomy, retroperitoneal lymphadenectomy, bladder neck surgery, pelvic surgery, spinal cord surgery, vasectomy. |
| History of mumps orchitis within three years of screening. |
| History of cryptorchidism |
| Dysorgasmia |
| Known or suspected cases of Klinefelter's syndrome or Kartagener's syndrome. |
| Clinical suspicion of varicocele |
| Any other condition that may affect the evaluation of the study. |
Table 1b: Exclusion criteria.
Interventions
SemenaxTM (capsules) is a proprietary dietary supplement containing various herbs, vitamin E, amino acids, and zinc. Its detailed composition is given in Table 2 (Table 2).
| Composition per dose | Zinc aspartate-30 mg, Vitamin-E-120.00 IU, L-carnitine-500 mg, Lepidium meyenii (Maca)-400 mg, Pinus pinaster (Pine) bark extract-300 mg, L-arginine HCl-250 mg, L-lysine-250 mg, Catuaba bark-200 mg, Epimedium sagitattum-150 mg, Muira puama-100 mg, Crataegus monogyna (Hawthorn)-50 mg, Cranberry extract-50 mg, Tribulus terrestris-50 mg, Avena sativa extract-50 mg, Smilax spp. (Sarsaparilla)-50 mg, Swedish Flower Pollen-50 mg, Cucurbita pepo (Pumpkin) seed-30 mg, and Butea superba-500 mg |
| Dosage | A total of 3200 mg of ingredients (equivalent to four capsules available commercially) |
| Regimen | 4 capsules two times a day orally |
| Batch number | T-F11040001 |
| Name and address of the manufacturer | Adroit pharmaceuticals Pvt. Ltd., |
| 46, Garoba Maidan, Itwari, Nagpur-440002, India | |
Table 2: Dietary supplement details.
Matching placebo capsules were prepared using microcrystalline cellulose. The supplement has already been marketed with a recommended daily dosage of 4 capsules, equivalent to the dose used for evaluation in the present study.
Study conduct
Participants were screened for the eligibility criteria defined for the study over the three screening visits. The eligible participants were then randomized to the study groups and provided with the IP. Subsequently, monthly follow-up study visits were conducted on day 30 and day 60 followed by a placebo run-out period of 15 days-20 days. The assessments done on each visit are described in Figure 1. At the end of the placebo run-out period again the semen analysis was performed. Treatment compliance was ensured by recording dispensed and returned medication at each visit. In addition, consumption of any concomitant medication was documented in the case report form.
Outcome measures
Efficacy outcomes: The objective of the study was to evaluate the effect of the investigational product on ejaculate volume, sperm count, sperm motility, and sperm morphology. The semen analysis was done as per WHO guidelines [2]. The participants abstained from sexual intercourse for a period of 2 days to 3 days each time before producing semen samples and complied with other semen collection procedures as recommended in the WHO laboratory manual. The number of responders for ejaculate volume improvement, i.e., individuals showing a 20% increase in ejaculate volume, was calculated.
The impact of the supplement on erectile function was measured using IIEF-EF and the total IIEF scores. The orgasm quality was assessed on the following scale: Grade 1-weak or poor, Grade 2-moderate or fair, Grade 3-good or strong, Grade 4-very good or very strong, Grade 5-most powerful or excellent. The responders for orgasm quality were defined as those participants who report one or more than one grade improvement in their orgasm quality during the study period. In addition, participants and the investigators rated the efficacy of the investigational product at the end of the study period using global assessment questionnaires.
The investigators performed a global assessment of efficacy based on the improvement in ejaculate volume, IIEF scores, and orgasmic quality as-excellent: Improvement in semen volume, IIEF scores, and orgasm quality, very good: Improvement in semen volume and IIEF scores or orgasm quality, good: Improvement in semen volume but no improvement in IIEF scores or orgasm quality, fair: No improvement in semen volume, but improved IIEF scores or orgasm quality, and poor: No improvement in any of the above parameters. The participant’s global assessment questionnaire included two items: “Did the treatment improve your ejaculate volume?’’ and “Did the treatment improve your orgasm quality?” with a response as “Yes” or “No”.
Safety outcomes: The safety outcome measures in the study included participants' rating on the tolerability assessment questionnaire as “good”, “fair”, or “poor” clinical and laboratory Adverse Events (AE) elicited from the change in Electrocardiogram (ECG), vitals (pulse rate and blood pressure), and laboratory parameters including Complete Blood Count (CBC), Erythrocyte Sedimentation Rate (ESR), Serum Glutamic Pyruvic Transaminase (SGPT), serum creatinine, and routine urine examination.
The 12-lead ECG of all the participants was performed in a resting state by the same technician and the report was prepared by a cardiologist. The pulse rate was measured manually by counting the number of beats per minute. The blood pressure was measured by a calibrated mercury sphygmomanometer by the investigator. CBC was analyzed using EDTA whole blood by a fully automated differential cell counter (Beckman Coulter AcT 5diff). ESR was determined using the Westergren method. SGPT (NADH (P-5’-P) method) and serum creatinine (alkaline picrate method) were analyzed using the ARCHITECT ci8200 System.
Ethical considerations
This study complied with the ethical guidelines of the “world medical association declaration of Helsinki-Ethical principles for medical research involving human subjects" adopted by the 18th WMA general assembly, Helsinki, Finland, June 1964 and amended by the 59th WMA general assembly, Seoul, South Korea, October 2008, International Conference On Harmonization-Good Clinical Practices (ICH-GCP), applicable regulatory guidelines, and ethical guidelines for biomedical research on human participants 2006, issued by the Indian Council of Medical Research (ICMR), India. In addition, the study protocol was approved by meet ethics committee, Mumbai, India. The trial was registered with a clinical trial registry, India (Reg. No. CTRI/2011/09/001995). The study aims, procedures, risks, and benefits involved were explained in vernacular language by the researchers to all the participants. Written consent was obtained from all the participants before recruitment into the study. The study report has been presented as recommended in the CONSORT statement.
Statistical analysis
As this was the first study of this supplement, therefore, based on previous similar studies [11,12] a minimum sample size of 60 evaluable participants, with 30 in each group, was considered appropriate to detect a statistically significant difference between the supplement and placebo. The efficacy analyses were done on the Per-Protocol (PP) population comprising participants reporting for all protocol stipulated study visits and did not have any major protocol deviations related to the primary efficacy outcome while the safety analyses were performed on the Intention-To-Treat (ITT) population
Descriptive statistics are presented as absolute counts, percentages, mean, and Standard Deviation (SD). Continuous baseline characteristics, efficacy, and safety parameters were compared using Analysis of Variance (ANOVA). Subgroup analysis was performed for two sub-groups: Normospermic and hypospermic. Pearson's Chi-square test was applied to analyze the categorical parameters such as grade of orgasms, number of responders, and participant's global efficacy and tolerability assessment, as well as investigator's global assessment. All statistical tests were performed at a 5% level of significance.
Quality Assurance
Several steps were taken to ensure the collection of accurate, consistent, complete, and reliable data. Before initiating the study, investigators and the study team were trained on the protocol, case report form filling rules, and administration of the study questionnaire to ensure appropriate and standardized data capture was organized. Site monitoring and audit were performed as per the pre-approved plan. Semen analysis was done as per the WHO recommendations, and all the laboratory technicians were trained to ensure uniformity during sample collection and analysis.
Results
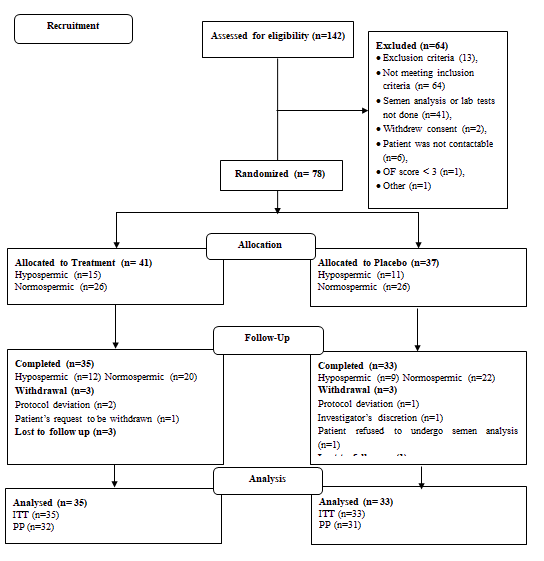
A total of 142 study participants were screened for the study, of which 64 were screening failures. The remaining 78 study participants were randomized to receive either the supplement (n=41) or placebo (n=37). A comparable number of hypospermic and normospermic men were included in both groups for subgroup analysis. The detailed participant disposition is provided in Figure 2 (Figure 2).
Demographic and baseline characteristics of participants
The demographic and baseline characteristics of both groups are presented in Table 3. The two groups and the subgroups based on semen volume were comparable for demographic and key baseline characteristics (Table 3).
| Baseline characteristics | Supplement (n=41) | Placebo (n=37) | ||
|---|---|---|---|---|
| Hypospermic (n=15) | Normospermic (n=26) | Hypospermic (n=11) | Normospermic | |
| (n=26) | ||||
| Age (years) | 38.60 (9.03) | 37.96 (6.60) | 36.73 (8.24) | 35.88 (6.73) |
| Pre-existing conditions | 0 | 3 | 0 | 4 |
| (Hypertension, peptic disease, URTI, hyperacidity) | ||||
| (Hyperacidity, psoriasis, joint pain) | ||||
| Concomitant medication | 0 | 2 | 0 | 2 |
| BMI (kg/m2) | 25.05 (4.13) | 24.08 (3.48) | 25.66 (2.88) | 24.91 (3.60) |
| Ejaculatory volume (ml) | 1.36 (0.39) | 3.22 (0.89) | 1.38 (0.34) | 3.08 (0.77) |
| Total IIEF score | 43.40 (8.97) | 43.62 (8.38) | 45.18 (7.37) | 43.92 (7.44) |
Table 3: Demographic and baseline characteristics of PP population. Data are expressed as Mean (SD) / n / n (%). BMI: Body Mass Index; IIEF: International Index Of Erectile Function; URTI: Upper Respiratory Tract Infection.
Efficacy outcomes
Ejaculate volume: The mean change in the ejaculatory volume from baseline to endline for both groups has been presented in Table 4. At the endline, there was an increase in the ejaculate volume (19.67%) in the supplement group from baseline values, whereas the placebo group showed a slight reduction (8%) in ejaculate volume. This change was statistically significant when compared across the two groups (p<0.05) (Table 4).
| Time points | Supplement (n=32) | Placebo (n=31) | p-value |
|---|---|---|---|
| Baseline (Day 0) | 2.49 (1.14) | 2.64 (1.00) | |
| Endline (Day 60) | 2.97 (1.44) | 2.43 (1.13) | |
| Change from baseline to endline | 0.49 (0.82) | -0.21 (0.75) | 0.0008 |
| p valueb | 0.14 | 0.44 |
Table 4: Effect of the IP on ejaculate volume (ml) of total PP population. Data are expressed as Mean (SD). a: ANOVA; b: Paired t-test.
Subgroup analyses conducted on hypospermic and normospermic populations have been presented in Table 4. There was an increase in ejaculate volume from baseline to endline in the hypospermic (33%) and normospermic (15%) subgroups within the supplement group, but this increase was not statistically significant (p>0.05). However, compared with the placebo group, the supplement group showed a statistically significant improvement in ejaculate volume (p<0.05) in the normospermic group only at the end of the study. On the other hand, the ejaculate volume of the placebo group showed no statistically significant change in both hypospermic and normospermic subgroups (Table 5).
| Hypospermic subgroup | |||
|---|---|---|---|
| Time points | Supplement (n=12) | Placebo (n=9) | p-value |
| Baseline (Day 0) | 1.32 (0.39) | 1.50 (0.23) | 0.24 |
| End line (Day 60) | 1.77 (0.87) | 1.50 (0.67) | 0.46 |
| Change from baseline to end line | 0.44 (0.81) | 0.00 (0.74) | 0.21 |
| p-valueb | 0.12 | 0.99 | |
| Normospermic subgroup | |||
| Time points | Supplement (n=20) | Placebo (n=22) | p-valuea |
| Baseline (Day 0) | 3.19 (0.80) | 3.11 (0.79) | 0.77 |
| End line (Day 60) | 3.70 (1.21) | 2.82 (1.06) | 0.02 |
| Change from baseline to end line | 0.51 (0.85) | -0.30 (0.75) | 0.002 |
| p-value | 0.12 | 0.3 | |
Table 5: Subgroup analysis of the effect of the IP on ejaculate volume (ml).
Number of responders
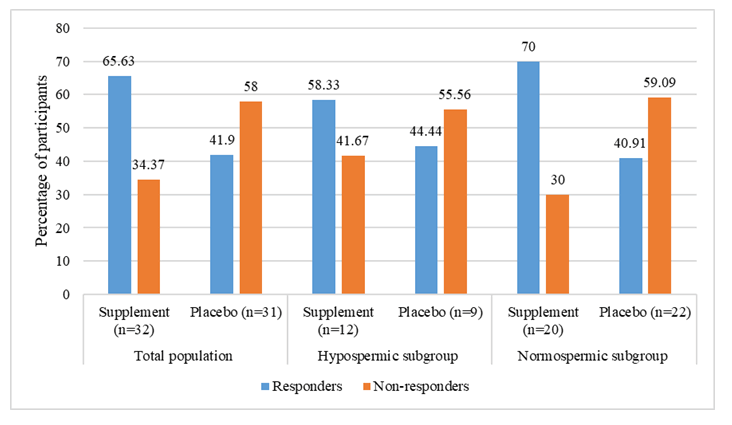
Participants showing a 20% or greater increase in the ejaculate volume from baseline to endline have been referred to as "responders," and those with an increase in ejaculate volume lower than the 20% threshold have been referred to as "non-responders”. The number of responders was higher in the supplement group (50%) than in the placebo group (16%), with a statistically significant difference being reported between the two study groups (p<0.05). In the hypospermic subgroup, though responders were higher in the supplement (58.33%) than in the placebo group (41.67%), the difference was not statistically significant (p=0.26). On the contrary, a statistically significant number of participants (45%) in the normospermic subgroup showed a 20% increase in ejaculate volume in the supplement group compared to the placebo group. However, the normospermic group had more non-responders (55% and 91%) in both groups (Table 6).
| Outcomes | Total PP population | p-valuea | |
|---|---|---|---|
| Supplement (n=32) | Placebo (n=31) | ||
| Responders | 16 (50) | 5 (16.13) | 0.004 |
| Non-responders | 16 (50) | 26 (83.87) | |
| Hypospermic subgroup | |||
| Supplement (n=12) | Placebo (n=9) | ||
| Responders | 7 (58.33) | 3 (25) | 0.26 |
| Non-responders | 5 (41.67) | 6 (75) | |
| Normospermic subgroup | |||
| Supplement (n=20) | Placebo (n=22) | ||
| Responders | 9 (45) | 2 (9) | 0.01 |
| Non-responders | 11 (55) | 20 (91) | |
Table 6: Responders analysis. Data are expressed as n (%). a:Chi-square test; PP: Per-Protocol.
Sperm characteristics
Table 6 presents the mean change in sperm characteristics-counts, motility, and morphology from baseline to endline. At the end of the study period, the sperm count in the supplement group improved significantly (11.67%) from the baseline values while it decreased (6.23%) in the placebo group. In addition, at the endline, the difference between the two groups was statistically significant (p<0.05). However, the change from baseline was statistically insignificant within and across the groups. In addition, progressive sperm motility showed a decreasing trend while non-progressive sperm motility showed an increasing trend in both groups without any statistical significance. Finally, sperm morphology improved slightly in the supplement group at the endline while it decreased in the placebo group, and this change was statistically insignificant within and between the two groups (Table 7).
| Sperm parameters | Time points | Supplement (n=32) | Placebo (n=31) | p-value |
|---|---|---|---|---|
| Sperm count (million/ml) | Baseline | 64.91 (46.55) | 52.46 (37.26) | 0.25 |
| End line | 72.48 (41.02) | 49.19 (41.18) | 0.03 | |
| Change from baseline to end line | 7.58 (31.47) | -3.27 (27.49) | 0.15 | |
| p-valueb | 0.49 | 0.74 | ||
| Progressive motility (%) | Baseline | 58.13 (20.01) | 55.29 (17.38) | 0.55 |
| End line | 55.27 (17.39) | 49.84 (15.44) | 0.2 | |
| Change from baseline to end line | -2.86 (16.45) | -5.45 (12.73) | 0.49 | |
| p-valueb | 0.54 | 0.2 | ||
| Non-progressive motility (%) | Baseline | 8.13 (6.63) | 8.82 (5.34) | 0.65 |
| End line | 10.20 (8.33) | 9.27 (5.63) | 0.61 | |
| Change from baseline to end line | 2.08 (3.73) | 0.45 (3.44) | 0.08 | |
| p-valueb | 0.27 | 0.75 | ||
| Sperm morphology (%) | Baseline | 82.02 (21.77) | 88.23 (15.42) | 0.2 |
| Endline | 86.05 (13.65) | 86.19 (15.31) | 0.97 | |
| Change from baseline to end line | 4.03 (18.06) | -2.03 (6.58) | 0.08 | |
| p-valueb | 0.38 | 0.6 |
Table 7: Effect of IP on sperm parameters in total PP population. Data are expressed as Mean (SD). a: ANOVA; b: paired t-test.
Table 8 presents the analyses performed on the hypospermic and normospermic subgroups, respectively. Sperm counts in the hypospermic subgroup within both groups increased compared with baseline. The absolute change was more in the supplement group than the placebo but was not statistically significant. In the same subgroup, a reduction in progressive and an increase in non-progressive sperm motility was observed, from baseline to endline, within the groups. However, in this regard, it is essential to note that the reduction was neither clinically significant nor statistically relevant. The hypospermic subgroup demonstrated a statistically significant difference between the two groups for sperm morphology (p=0.05), where the normal forms increased in the supplement group and decreased in the placebo group.
The analysis performed on the normospermic subgroup revealed a few crucial differences from baseline to endline. First, the placebo group witnessed a reduction in sperm count from baseline to endline, whereas an increase was seen in the supplement group. Second, the progressive motility was reduced in both the groups at the endline; however, this reduction lacked statistical significance. On the other hand, non-progressive motility improved at the end of the study in both groups. This change was statistically significant across the group. Finally, in the placebo group, a marginal reduction was observed, from baseline to endline, in the number of sperms with normal morphology. The number of morphologically normal sperms increased, from baseline to endline, in the supplement group. However, the difference between the two groups for sperm morphology was not statistically significant.
| Sperm parameters | Timepoints | Hypospermic subgroup | Normopermic subgroup | ||
|---|---|---|---|---|---|
| Supplement (n=12) | Placebo (n=9) | Supplement (n=20) | Placebo (n=22) | ||
| Sperm count | Baseline | 85.26 (58.66) | 53.66 (35.66) | 52.69 (33.54) | 51.97 (38.71) |
| (million/ml) | End line | 89.28 (47.80) | 53.78 (40.34) | 62.41 (33.72) | 47.32 (42.31) |
| Change from baseline to end line | 4.02 (24.59) | 0.12 (26.20) | 9.71 (35.40) | -4.65 (28.48) | |
| Progressive motility (%) | Baseline | 56.67 (21.38) | 51.28 (18.54) | 59.00 (19.66) | 56.93 (17.06) |
| End line | 53.13 (17.65) | 43.33 (18.75) | 56.55 (17.57) | 52.50 (13.45) | |
| Change from baseline to end line | -3.54 (12.54) | -7.94 (10.06) | -2.45 (18.70) | -4.43 (13.76) | |
| Non-progressive motility (%) | Baseline | 10.42 (8.65) | 11.50 (7.62) | 6.75 (4.80) | 7.23 (3.77) |
| End line | 12.08 (9.40) | 12.50 (8.29) | 9.08 (7.65) | 7.95 (3.59) | |
| Change from baseline to end line | 1.67 (3.89) | 1.00 (4.36) | 2.33 (3.71) | 0.23 (3.08) | |
| Sperm morphology (%) | Baseline | 83.17 (14.31) | 86.17 (21.10) | 81.32 (25.56) | 89.07 (12.94) |
| End line | 84.42 (13.62) | 81.44 (18.32) | 87.03 (13.92) | 88.14 (13.91) | |
| Change from baseline to end line | 1.25 (4.96) | -4.72 (8.45) | 5.70 (22.59) | -0.93 (5.51) | |
Table 8: Subgroup analysis of the effect of IP on sperm parameters. Data are expressed as Mean (SD).
IIEF-total and erectile function subscale scores
Table 8 presents the mean change from baseline to endline of total IIEF and IIEF-EF scores for the total PP population. Within-group total IIEF score for supplement and placebo groups was increased to statistically significant levels at the end of the study, and the increase was more in the supplement group (17.43% vs. 11.81%). However, this change was statistically comparable between the two groups. Similar observations were made for the erectile function domain score of IIEF (19.08% vs. 13.54%).
| Parameters | Timepoints | Total PP population | p-valuea | |
|---|---|---|---|---|
| Supplement (n=32) | Placebo (n=31) | |||
| Total IIEF score | Baseline | 43.37 (8.81) | 43.42 (7.18) | 0.98 |
| End line | 50.94 (9.41) | 48.55 (7.31) | 0.27 | |
| Change from baseline to end line | 7.56 (5.64) | 5.13 (7.36) | 0.15 | |
| p-valueb | 0.002 | 0.01 | ||
| IIEF-EF score | Baseline | 17.19 (4.04) | 17.65 (3.19) | 0.62 |
| End line | 20.47 (3.58) | 20.03 (3.18) | 0.61 | |
| Change from baseline to end line | 3.28 (3.03) | 2.39 (3.52) | 0.28 | |
| p-valueb | 0.001 | 0.005 | ||
| Hypospermic subgroup | ||||
| Parameters | Time points | Supplement (n=12) | Placebo (n=9) | p-valuea |
| Total IIEF score | Baseline | 43.58 (9.62) | 45.11 (7.34) | 0.7 |
| End line | 49.58 (9.05) | 48.44 (4.61) | 0.73 | |
| Change from baseline to end line | 6.00 (5.08) | 3.33 (6.67) | 0.31 | |
| p-valueb | 0.13 | 0.26 | ||
| IIEF-EF score | Baseline | 17.08 (4.48) | 17.89 (4.40) | 0.69 |
| End line | 19.67 (3.84) | 20.22 (2.54) | 0.71 | |
| Change from baseline to end line | 2.58 (2.50) | 2.33 (3.77) | 0.86 | |
| p-valueb | 0.14 | 0.19 | ||
| Normospermic subgroup | ||||
| Parameters | Time points | Supplement (n=20) | Placebo (n=22) | p-valuea |
| Total IIEF score | Baseline | 43.25 (8.54) | 42.73 (7.17) | 0.83 |
| End line | 51.75 (9.77) | 48.59 (8.26) | 0.26 | |
| Change from baseline to end line | 8.50 (5.87) | 5.86 (7.64) | 0.22 | |
| p-valueb | 0.006 | 0.01 | ||
| IIEF-EF score | Baseline | 17.25 (3.88) | 17.55 (2.67) | 0.77 |
| End line | 20.95 (3.43) | 19.95 (3.46) | 0.35 | |
| Change from baseline to end line | 3.70 (3.29) | 2.41 (3.50) | 0.23 | |
| p-valueb | 0.003 | 0.01 | ||
Table 9: Effect of IP on IIEF and IIEF-EF scores. Data are expressed as Mean (SD); a: ANOVA; b: Paired t-test; IIEF-EF: International Index of Erectile Function-Erectile Function.
On subgroup analysis (Table 8), it was observed that there was no statistically significant change in the total or EF domain scores in the hypospermic population though a positive trend was noticed in both the groups at the end of the study period. This finding was in contrast to those observed in the normospermic population wherein there was a statistically significant improvement in total and IIEF-EF scores in both the supplement and the placebo group from baseline to endline, which was comparable in the two groups.
Orgasm quality
Participants were asked to grade their orgasm quality at baseline and end line on a 5-point scale. Participants with an increase of one or more points on the grade scale were considered "responders" whereas participants who did not show any change on this scale were referred to as "non-responders". Overall there were 34 responders. The responders were higher in the supplement group (n=21) than placebo (n=13), though both groups were statistically comparable. Subgroup analysis also showed the same trend (Figure 3).
Investigators' global assessment
Based on the improvement in ejaculate volume, IIEF scores, and orgasmic quality, investigators rated the supplement's efficacy at the endline on a 5-point scale: Excellent, very good, good, fair, and poor. There was a statistically significant difference between the two groups (p<0.05). More than 65% of participants in the supplement group rated the product “good” and above compared to 61% in the placebo group.
Participants' global efficacy assessment
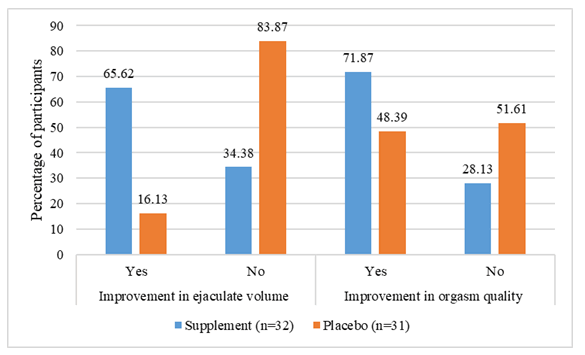
Study participants were asked to assess the intervention’s efficacy based on perceived ejaculate volume and orgasmic quality improvement. The number of participants with perceived improvement in ejaculate volume (65.62% vs. 16.13%) and orgasm quality (71.87% vs. 48.39%) was higher in the supplement group than placebo (Figure 4). Furthermore, improvement in ejaculate volume was statistically significant (p<0.05), whereas improvement in orgasmic quality just missed reaching statistical significance (p>0.05).
On subgroup analysis, the hypospermic subgroup perceived a statistically significant improvement in ejaculate volume (yes: 7 vs. 1; no: 5 vs. 8; p=0.03) in the supplement arm as compared with placebo. On the other hand, no statistically significant difference (yes: 8 vs. 5; no: 4 vs. 4; p=0.60) was noted between the groups for orgasm quality. In the normospermic subgroup, supplement group demonstrated statistical significance for both these parameters over placebo (ejaculate volume-yes: 14 vs. 4; no: 6 vs. 18; p=0.001, orgasmic quality-yes: 15 vs. 10; no: 5 vs. 12; p=0.05).
Safety evaluations
No clinically relevant or statistically significant changes were observed in any of the vitals or laboratory parameters from baseline to endline, either within or across the two groups. The majority of study participants rated their tolerability of the IP as 'good' in both groups (86.84% & 88.57%) on the tolerability rating scale. Hence the two groups were comparable (p=0.82). In addition, none of the participants reported tolerability as 'poor' in both groups.
Adverse events
A total of 15 AEs were reported during the study, which were either mild (n=8) or moderate (n=7) in intensity. Five AEs (hyperacidity-2, abdominal pain-2, and stomach bloating-1) were probably related to the IP, one (headache) was possibly related, and 9 AEs were unrelated to the IP. All AEs resolved during the study without any sequelae. During the study, there were no deaths, serious adverse events, or other significant AEs. In addition, there were no abnormal ECG findings at the endline.
Discussion
The current study was an exploratory, randomized, double-blind, and placebo-controlled clinical investigation to assess the safety and efficacy of the dietary supplement in men desiring improvement in semen quality. The study results showed that the supplement could improve the ejaculate volume significantly after two months of administration compared to the placebo. On subgroup analysis, it was revealed that this effect was more pronounced in the healthy normospermic population. On the other hand, in the hypospermic population, it could not reach the significance level.
The supplement group showed more responders than the placebo group, with the difference between the two groups being statistically significant. Again the efficacy of the supplement was more in the normospermic subgroup. For sperm characteristics, the hypospermic group performed statistically better in improving the morphology, while the normospermic group showed improvement in non-progressive motility of the sperms.
The supplement and placebo both improved the total IIEF score and IIEF-EF score significantly from the baseline scores. In addition, the number of participants with an increase in grade of orgasm intensity was comparatively higher in the treated group than placebo. Analysis of both participants' and investigators' global assessment of therapy demonstrated a statistically significant advantage obtained with the use of supplement over placebo. In the overall appraisal of efficacy results, the supplement was more efficacious than the placebo in increasing ejaculate volume and improving sexual function.
Participants tolerated the supplement well during the study. All AEs were mild to moderate in intensity and were successfully resolved during the study. None of the laboratory parameters, ECG, and vital signs showed clinically relevant or statistically significant change from baseline to endline. Participants' tolerability was also rated as 'good' by most of them. Previous published preclinical studies have shown the efficacy of polyherbal mixtures in managing male sexual dysfunction [10]. In clinical studies also, herbal formulations have shown great promise in improving ejaculate volume and sperm characteristics. Kolahdooz et al. reported an 11.67% increase in semen volume after a daily intake of 5 ml of Nigella sativa oil for two months [13]. The current study has shown a 19.68% increase in the ejaculate volume in the supplement group. Several herbs have been traditionally used and acknowledged for their role in male sexual dysfunction and impotence [14,15]. Many of the ingredients of the dietary supplement are also traditionally known to improve male sexual performance, ejaculate volume, and fertility. Few clinical studies have shown the efficacy of the individual components of the supplement, such as Maca [16]. L-carnitine, L-arginine [8], and Zinc [7], in improving seminal characteristics. However, this was the first study to provide preliminary clinical evidence supporting the claims of the polyherbal dietary supplement. The supplement has shown promising efficacy in increasing the ejaculate volume over placebo in a short duration of two months. To detect a change in sperm parameters and IIEF scores, it is recommended to conduct a study for an extended period and on larger sample size.
Conclusion
Overall, the polyherbal dietary supplement has demonstrated efficacy in improving the ejaculate volume and orgasm quality to statistically significant levels in the study participants. It has demonstrated a good acceptability and safety profile. The study opens new avenues for men desiring to improve their semen quantity and sperm quality with better orgasm.
About the authors
•Doug Mackay is the chief operating officer at leading edge health, Canada. He has approved the manuscript.
•Sonika Verma is senior manager–science and communications at Vedic life sciences Pvt. Ltd., Mumbai and has written the manuscript.
•Shalini Srivastava is associate director-clinical development at Vedic life sciences Pvt. Ltd., Mumbai. She has edited and approved the manuscript.
Funding
The study was funded by leading edge health, Victoria, Canada.
Conflict of interest
Doug Mackay is affiliated with leading edge health, Victoria Canada. The other authors declare no conflict of interest.
Acknowledgments
The authors are grateful to all the participants who volunteered for the study and the investigators who executed the study. Further, the authors are thankful to the clinical research organization Vedic life sciences Pvt. Ltd., Mumbai, India for facilitating the study and leading edge health, Victoria, Canada, for providing the study products.
References
- Bjorndahl L, Kvist U (2003) Sequence of ejaculation affects the spermatozoon as a carrier and its message. Reprod Biomed Online 7: 440–448.
[Crossref], [Google Scholar], [Indexed]
- https://www.who.int/docs/default-source/reproductive-health/srhr-documents/infertility/examination-and-processing-of-human-semen-5ed-eng.pdf
- Owen DH, Katz DF (2005) A review of the physical and chemical properties of human semen and the formulation of a semen simulant. J Androl 26: 459–469.
[Crossref], [Google Scholar], [Indexed]
- Roberts M & Jarvi K (2009) Steps in the investigation and management of low semen volume in the infertile man. Can Urol Assoc J 3: 479–485.
[Crossref], [Google Scholar], [Indexed]
- https://urology.ucsf.edu/patient-care/adult-non-cancer/male-sexual-and-reproductive-health/ejaculatory-orgasmic-disorders#.Yk_ZFMhBzIV
- Nouri M, Amani R, Nasr-Esfahani M, Tarrahi MJ (2019) The effects of lycopene supplement on the spermatogram and seminal oxidative stress in infertile men: A randomized, double-blind, placebo-controlled clinical trial. Phytother Res 33: 3203–3211.
[Crossref], [Google Scholar], [Indexed]
- Zhao J, Dong X, Hu X, Long Z, Wang L, Liu Q, et al. (2016) Zinc levels in seminal plasma and their correlation with male infertility: A systematic review and meta-analysis. Sci Rep 6: 22386.
[Crossref], [Google Scholar], [Indexed]
- Stanislavov R, Rohdewald P (2014) Sperm quality in men is improved by supplementation with a combination of L-arginine, L-citrullin, roburins and Pycnogenol®. Int Urol Nephrol 66: 217–223.
[Crossref], [Google Scholar], [Indexed]
- Lipovac M, Bodner F, Imhof M, Chedraui P (2016) Comparison of the effect of a combination of eight micronutrients versus a standard mono preparation on sperm parameters. Reprod Biol Endocrinol 14: 84.
[Crossref], [Google Scholar], [Indexed]
- Hussain SA, Hameed A, Nasir F, Wu Y, Suleria H, Song Y (2018) Evaluation of the spermatogenic activity of polyherbal formulation in oligospermic males. BioMed Res Int: 2070895.
[Crossref], [Google Scholar], [Indexed]
- Rolf C, Cooper TG, Yeung CH, Nieschlag E (1999) Antioxidant treatment of patients with asthenozoospermia or moderate oligoasthenozoospermia with high-dose vitamin C and vitamin E: A randomized, placebo-controlled, double-blind study. Hum Reprod 14: 1028-1033.
[Crossref], [Google Scholar], [Indexed]
- Balercia G, Buldreghini E, Vignini A, Tiano L, Paggi F, Amoroso S, et al. (2009) Coenzyme Q10 treatment in infertile men with idiopathic asthenozoospermia: A placebo-controlled, double-blind randomized trial. Fertil Steril 91: 1785–1792.
[Crossref], [Google Scholar], [Indexed]
- Kohladooz M, Nasri S, Modaress SZ, Kianbakht S, Huseini HF (2014) Effects of Nigella sativa L. seed oil on abnormal semen quality in infertile men: A randomized, double-blind, placebo-controlled clinical trial. Phytomedicine 21: 901-905.
[Croosref], [Google Scholar], [Indexed]
- Jaradat N, Zaid AN (2019) Herbal remedies used for the treatment of infertility in males and females by traditional healers in the rural areas of the west bank/Palestine. BMC Complement Altern Med 19: 194.
[Crossref], [Google Scholar], [Indexed]
- Abarikwu SO, Onuah CL, Singh SK (2020) Plants in the management of male infertility. Andrologia 52: e13509.
[Crossref], [Google Scholar], [Indexed]
- Melnikovova I, Fait T, Kolarova M, Fernandez EC, Milella L (2015) Effect of lepidium meyenii walp on semen parameters and serum hormone levels in healthy adult men: A double-blind, randomized, placebo-controlled pilot study. Evid Based Complement Altern Med: 324369.
[Crossref], [Google Scholar], [Indexed]
Open Access Journals
- Aquaculture & Veterinary Science
- Chemistry & Chemical Sciences
- Clinical Sciences
- Engineering
- General Science
- Genetics & Molecular Biology
- Health Care & Nursing
- Immunology & Microbiology
- Materials Science
- Mathematics & Physics
- Medical Sciences
- Neurology & Psychiatry
- Oncology & Cancer Science
- Pharmaceutical Sciences