Role of Peroxidase in Clinical Assays: A Short Review
Anantharaman Shivakumar, Jashmitha BG and Dhruvaraj MR
DOI10.4172/2472-1921.100048
PG Department of Chemistry, St. Philomena’s College, Mysore-570015, Karnataka, India
- *Corresponding Author:
- Dr. Shivakumar A
Assistant Professor
PG Department of Chemistry
St. Philomena’s College
Bangalore-Mysore Road, Bannimantap
Mysore-570 015, Karnataka, India
Tel: 0821 424 0900
E-mail: shivakem77@yahoo.co.in
Received Date: May 03, 2017; Accepted Date: May 09, 2017; Published Date: May 12, 2017
Citation: Shivakumar A, Jashmitha BG, Dhruvaraj MR. Role of Peroxidase in Clinical Assays: A Short Review. J Clin Nutr Diet. 2017, 3:2. doi: 10.4172/2472-1921.100048
Abstract
Development of sensitive enzymatic methods for hydrogen peroxide is key for the quantification of several bioconstituents such as glucose, triglycerides, creatinine, and uric acid and so on. Hydrogen peroxide released by the oxidase enzymes are quantified by peroxidase enzyme involving spectrophotometry, fluorimetry, chemiluminisence, potentiometric sensing, amperometric, coulometric and such others. Authors in this line, present a short review on the assay of peroxidase. The entire review is divided into three different sections; first the importance of peroxidase clinically, secondly peroxidase chemistry with hydrogen peroxide and finally its role in the assay of bioconstituents.
Keywords
Peroxidase; Clinical importance; Oxidase enzymes; Glucose assay; Nano sensors
Peroxidase: A Clinically Important Enzyme
Peroxidases are widely distributed in nature especially in animal and plant cells. Peroxidases comprise of three major categories; plant peroxidases, animal peroxidases and catalases. These enzymes utilize hydrogen peroxide to catalyze the oxidation of variety of organic and inorganic compounds.
Over the years the development in clinical and diagnostic techniques, redox natured reactions are gaining vital importance. Biochemical reactions that are redox are gaining prominence in basic as well as applied research. Peroxidases are the driving force among these as most of the clinically important assays such as glucose; creatinine and uric acid determinations cannot be carried out without peroxidases. Immobilization of peroxidase enzyme has received much attention in the construction of biosensors due to economical and easy viability in enzyme immunoassays and enzyme linked immunosorbent assays. Keeping the views in logical frame work, the authors discuss the important role of peroxidase in clinically important assays, its chemistry behind hydrogen peroxide reaction extending to assay of diabetically important glucose and finally providing a tabulated physiological range of biochemical components in blood.
Peroxidase: A Friend to Oxidase Enzymes
The reactions catalyzed by peroxidases can be grouped into oxidative dehydrogenation, oxygen transfer, oxidative halogenations and hydrogen peroxide dismutation. Among these, oxidative dehydrogenation has greater application in analytical biochemistry.
Peroxidases are having great potential application as they can be used in a diagnostic kit for hydrogen peroxide, glucose and oxidase enzyme determination [1]. In particular, HRP are widely used in research areas such as enzymology, biochemistry, medicine, genetics, physiology, histo and cyto chemistry because of easy availability, economical and high catalytic activity [2]. The commercial production of peroxidase has increased due to its analytical diagnostics particularly biosensing, in immunosensors and in nucleic acid detection. Heme proteins named peroxisomes are present in high concentration in cell compartments preventing excessive accumulation of peroxide, a powerful oxidizing agent. HRP meets the entire requirements for successful analytical enzymology because of its specificity, flexibility in assay, stability, sensitivity of detection, and availability in pure form. HRP on coupling with oxidase enzyme can be used for analysis of wide range of analytes such as glucose, cholesterol, lactic acid, choline, xanthine, uric acid, bilirubin, and creatinine.
Combination of peroxidase and indole-3-acetic acid is currently being used as a cancer therapeutic agent [3]. The therapeutic procedure involves two steps; first decoding of enzyme in the tumor cells, second administering specific prodrug, indole-3- acetic acid which gets converted into cytotoxic drug by the enzyme expressed in the target tumor. The decarboxylated form of radical cation indole-3-acetic acid can conjugate with DNA and other biological nucleophiles [4]. Some other anticancer strategies adopted include antibody and polymer directed enzyme/prodrug therapy.
Chemistry of Reaction between Peroxidase and Hydrogen Peroxide
Peroxidases can catalyze a variety of organic substrates such as phenols, and aromatic amines with two step three intermediate cyclic processes. The process is as under
Step I: The formation of HRP-I with the incorporation of oxygen by the reaction with hydrogen peroxide (oxoferryl). The oxidation state of iron is +4 along with porphyrins-π-radical cation.
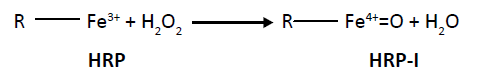 (1)
(1)
HRP HRP-I
Step II: This HRP-II has a tendency to oxidize organic substrates by single electron transfer mechanism
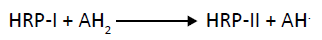 (2)
(2)
Step III: The conversion of HRP to native state by second substrate molecule.
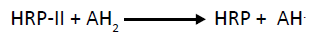 (3)
(3)
The structures of oxoferryl species as confirmed by different research groups.
The initial steps of the reaction between peroxidase and hydrogen peroxide involves the basic amino acid residue (B), in the reaction [5] is depicted as:
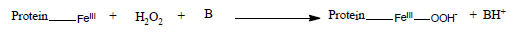 (4)
(4)
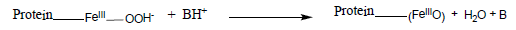 (5)
(5)
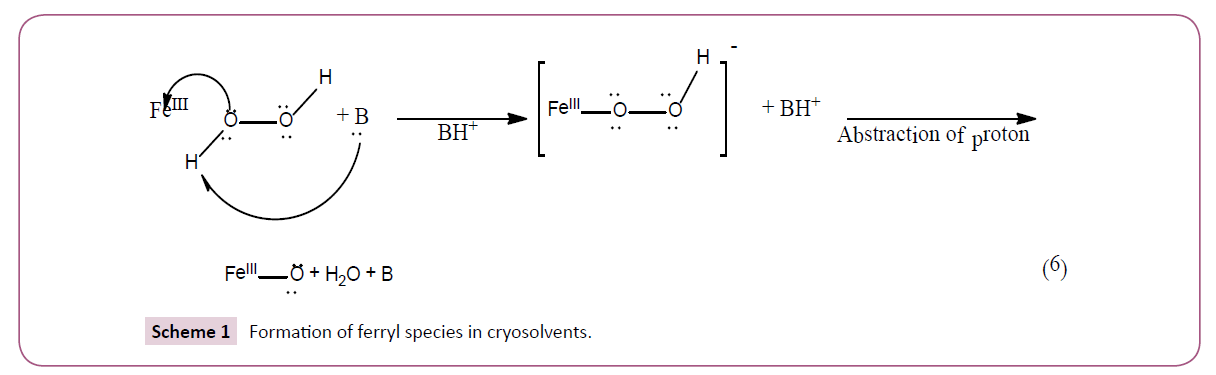
The intermediate of the reaction is highly unstable with spectrum has been reported in cry solvents at temperatures less than -160C [6,7] (Scheme 1).
The initial interaction between peroxidase and hydrogen peroxide consist the formation of two electron bonds between iron (III) and one of the peroxide oxygen’s. The basic amino acid residue acts as a mediator in the transfer of proton between α and β oxygen which subsequently reacts and loses a molecule of water along with the release of basic amino acid residue. The final oxy heme structure consists of either a double bond or a dative bond with both electrons originating from an oxygen atom. This ferryl intermediate is responsible for the formation of hydroxyl free radical which in turn causes tissue damage in vivo suggesting that the oxygen may dissociate easily from the heme protein.
The oxyhemoglobin model [5] includes linear, bent, ring, met-superoxide model and two electron oxidative additions as listed in Table 1.
| Model | Description of model |
|---|---|
| Linear | 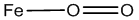 |
| Bent | 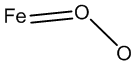 |
| Ring model | 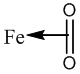 |
| Met-superoxide | 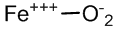 |
| Two electron oxidative | 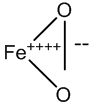 |
Table 1: Structures of oxyhemoglobin.
Glucose: An Often Difficult Species for Quantification
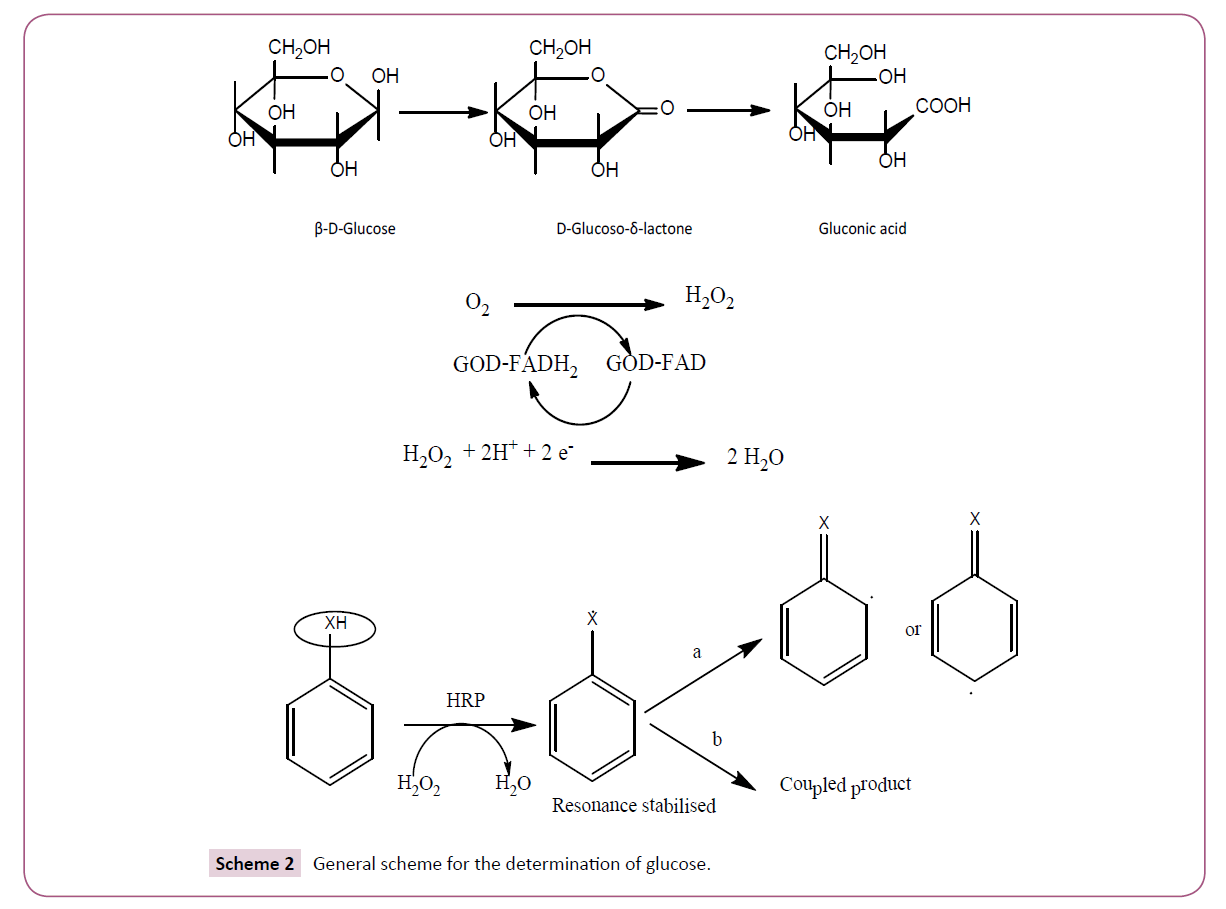
The two different structures glucose are closed loop and open chain. The closed loop α and β structures are in equilibrium with each other. The open chain structure has an aldehydic reactive group, which can undergoes oxidation resulting in the formation of gluconic acid with the concominant formation of hydrogen peroxide. The gluconic acids are difficult to quantify by the conventional colorimetric methods as carboxylic acids are least accessible for colorimetric methods. The blessing in disguise is the formation of hydrogen peroxide as a byproduct in the oxidation of glucose by glucose oxidase. The hydrogen peroxide liberated is active in various instrumental methods such as spectrophotometric, fluorimetric, potentiometric, amperometric and enzymatic approaches. Enzymes that use hydrogen peroxide as a substrate include catalase and peroxidase. Catalase decomposes hydrogen peroxide into water and oxygen. Oxygen can be determined by monometric or UV-Vis spectrophotometric or electrodic methods. Peroxidase converts hydrogen peroxide into a highly reactive hydroxyl radical, a powerful oxidant. Hydroxyl radical can oxidize various benzene centered species into a highly reactive radical intermediate which in turn can couple with secondary reagents such as phenol or amine to result in colorimetric signal. Enzymes that oxidize glucose as a principal substrate include glucose dehydrogenase, Quinoprotein glucose dehydrogenase, glucose-1-oxidase, and glucose-2-oxidase and glucose oxidase. Glucose oxidase is the only enzyme used in the oxidation of glucose because glucose dehydrogenase and quinoprotein glucose dehydrogenase are specific for β form of glucose with high turnover rates but the former is less soluble while the latter is unstable. Glucose-2- oxidase also oxidizes other carbohydrates such as xylose and glucolactone but lack of specificity overrules its use [8]. The glucose oxidase can be isolated from variety of sources among these Aspergillus niger is very often used. It is more stable in the physiological pH at which quantitative analysis can be carried out. The most commonly used substrates are o-dianisidine, tetremethylbenzidine, ABTS, 4-aminoantipyrine and phenol, Guaiacol, pyrogallol, cyanine dyes. Our research group has proposed different analytical probes for the assay of hydrogen peroxide involving HRP which include PPDD-DMAB, PPDD-NEDA and DMA [9]. Nano, a dwarf technology with one of its dimension measured in nanometer, are also not left behind in the analysis. Nanoparticles mimic the behavior of peroxidase enzyme, and they can be used as substitutes for peroxidase in the assay of glucose in the physiological range. Use of nanoparticles help to overcome certain important constraints like; control of temperature in enzymatic assay, involving several steps to immobilize the enzyme on the solid support thereby reducing the enzyme activity, waste of expensive enzymes because of its non-utilization completely, reduce the cost. Nanoparticles attracted scientific and technological interest because of ease of preparation, biocompatibility, and large surface to volume ratio collective oscillations of the surface electrons emitting light in the visible range. Table 2 lists some nanosensors used in glucose determination.
| Nano substrate | Linearity range | Reference |
|---|---|---|
| Silver nanoprism | 0.2-100 μM | [10] |
| Multiwalled carbon nanotube | 0-5 mM | [11] |
| CuS nanoparticle | 2-1800 μM | [12] |
| ZnFe2O4 | 1.25-18.75 μM | [13] |
| Mn doped ZnS Quantum dots | 10-0.1 mM and from 0.1-1 mM | [14] |
| Fe3O4 | 3.0-1000 μM | [15] |
| Cupric oxide | 0.1-8.0 mM | [16] |
| Carbon nitride dots | 1.0-1.0 mM | [17] |
| Prussian nanoparticles | 0.1-50.0 μM | [18] |
Table 2: Glucose linearity range of some nanosensors.
Glucose Monitoring Methods: Invasive and Non-Invasive
The normal physiological range of glucose is 3.3-6.6 mM (60 mg/ dL to 120 mg/dL). Frequent glucose determination in diabetic patients is required for the maintenance of glucose in blood. The invasive methods require regular lancing and finger bleeding. The noninvasive methods overcome pain in blood extraction, exposure to sharp objects, biohazard materials and the potential for increased frequency of testing [19]. The dual enzymatic colorimetric method involves glucose oxidase and peroxidase. The glucose oxidase catalyses the conversion of β-D-glucose by molecular oxygen producing gluconic acid and hydrogen peroxide. Glucose oxidase requires redox co-factor, FAD, which gets reduced to FADH2. The co-factor is regenerated by molecular oxygen with the formation of hydrogen peroxide. Furthermore, hydrogen peroxide can be monitored by spectrophotometry, fluorimetry, chemiluminisence, potentiometric sensing, amperometric, coulometric and such others
The spectrophotometric, fluorimetric and luminescence methods involve the formation of the colored or luminescence active product. In potentiometry, amperometry and coulometric, the electrons liberated/consumed is directly proportional to the number of glucose molecules as shown in Scheme 2.
Biosensor is in the modification process which was initially proposed by Clark and Lyon of Children hospital in Cincinnati. The glucose biosensor was composed of oxygen electrode coated with a thin layer of glucose oxidase and an outer dialysis membrane. Decrease in the concentration of oxygen is taken as a measure of glucose concentration in the blood or urine sample. The major disadvantage in this is operational potential for hydrogen peroxide and oxygen deficit in the samples. The high operating potential of biosensors was interfered by ascorbic acid, uric acid and drugs like acetoaminophen. These problems led to the development of second generation sensors such as ferrocene, ferricyanide, quinines, tetrathialfulvene, tetracyanoquinodimethane, thionine and methylene blue by the electron mediators to get improved performance. The use of mediators was overcome in the third generation biosensors, which have direct electron transfer reaction between the enzyme and the electrode.
Other non-invasive approaches include polarimetry [20] Raman spectroscopy [21], photoacoustics [22], optical coherence tomography [23], and photothermal deflectometer enhanced by total internal reflection [24].
As per the World Health Organization diagnosis should be ASSURED: Affordable, Sensitive, Specific, User friendly, Rapid and robust, Equipment free and Delivery to end users [25]. Point care of testing, microfluidic paper based analytical devices is rapid, less expensive, and more multiplexed than the instrumental analysis. They require small volume of liquid with little or no extra instrument for the quantification of biocomponent, a visual differentiation is only sufficient in making the near conclusion.
Enzyme immunoassay describes a broad range of assays to qualify and quantify antigens or antibodies. A typical assay involves immobilization of antigen with antibody on to a solid support. A secondary antibody labeled with peroxidase provides a detectable signal in the presence of colorimetric indicator. These are extensively used in enzyme linked immuno sorbent assays, western blotting and immuno-histochemistry techniques. Table 3 provides physiological range of bioconstituents in blood.
| Secondary enzyme | Substrate for secondary enzymes | Coenzyme/cofactor | Secondary product | Optimal pH | Normal physiological range of substrates* | References |
|---|---|---|---|---|---|---|
| Glucose oxidase | β-D-glucose | FAD | D-glucono-δ-lactone | 5.5 | 60-120 mg/dL | [27] |
| Alcohol oxidase | Primary alcohol | NAD+ | Aldehyde | 7.5 | - | [28] |
| Lactate oxidase | Lactic acid | FMN | Pyruvic acid | 7.2 | 4-20 mg/dL | [29] |
| Choline oxidase | choline | FAD | Betaine | 9-10 | - | [30] |
| D-aminoacid oxidase | D-aminoacid | FAD | α-ketoacid | - | 30-50 mg/dL | [31] |
| L-amino acid oxidase | L-aminoacid | FMN | 2-oxoacid | - | 30-50 mg/dL | [32] |
| Xanthine oxidase | Hypoxanthine | FAD | Uric acid | 7.5 | - | [33] |
| Uricase | Uric acid | Copper | Allantoin | 6.5-7.5 | 3.0-7.0 mg/dL | [34] |
| Cholesterol oxidase | Cholesterol | FAD | Cholest-4-en-3-one | 7.0-9.0 | 150- 200 mg/dL** | [35] |
| Polyamine oxidase | N1-acetylspermine | FAD | N1-acetylspermidine and 3-aminopropanal | 7.4 | - | [36] |
| Bilirubin oxidase | Bilirubin | - | biliverdin | 8.5 | 0.2-2.0 mg/dL** | [37] |
| Glutamate oxidase | L-glutamate | FAD | 2-oxoglutarate | 7 | 8.0-10.0 mg/dL | [37] |
Note: Optimum pH depends on the nature of substrates, co-substrates and methodology adopted in the determination
Table 3: Physiological range of bioconstituents in blood (*Reference values from clinical trials books; ** Values refer to total concentration).
Conclusion
Peroxidase has a prominent place in biotechnology and associated areas such as microbiology, histochemistry, medicine, genetics, and clinical chemistry due to its versatility, viability, and economical nature. The major problems with peroxidase enzyme are suicide killing with the concominant release of hydroxyl radical and lack of analysis at high temperature. Hence mutated peroxidase from different plant sources with improved stability and activity will be better contender for bioconstituents application. The use of nanomaterials which strongly influences the properties like stiffness, elasticity, and biocompatibility environment for oxidase enzymes can avoid peroxidase in the dual enzymatic systems.
Acknowledgements
One of the authors (A.S.K) expresses gratitude for the generous support by University Grants Commission, New Delhi, India (2166- MRP/15-16/KAMY006/UGC-SWRO). Also thank St. Philomena’s college for providing facilities to carry out the review.
References
- Song Y, Qu K, Zhao C, Ren J, Qu X (2010) Graphene Oxide: Intrinsic Peroxidase Catalytic Activity and Its Application to Glucose Detection. Adv Mater 22: 2206-2210.
- Sanz V, de Marcos S, Castillo J (2005) R, Galbán J. Application of Molecular Absorption Properties of Horseradish Peroxidase for Self-Indicating Enzymatic Interactions and Analytical Methods. J Am Chem Soc 127: 1038-1048.
- Veitch NC (2004) Horseradish peroxidase: a modern view of a classic enzyme. Phytochemistry 65: 249-259.
- Azevedo AM, Martins VC, Prazeres DMF, Vojinović V, Cabral JMS, et al. (2003) Horseradish peroxidase: a valuable tool in biotechnology. Biotechnol Ann Rev Elsevier 9: 199-247.
- Everse J (1998) The Structure of Heme Proteins Compounds I and II: Some Misconceptions. Free Radic Biol Med 24: 1338-1346.
- Baek HK, Van Wart HE (1992) Elementary steps in the reaction of horseradish peroxidase with several peroxides: kinetics and thermodynamics of formation of compound 0 and compound I. J Am Chem Soc 114: 718-725.
- Baek HK, Van Wart HE (1989) Elementary steps in the formation of horseradish peroxidase compound I: direct observation of compound 0 a new intermediate with a hyperporphyrin spectrum. Biochemistry 28: 5714-5719.
- Wilson R, Turner APF (1992) Glucose oxidase: an ideal enzyme. Biosensors and Bioelectronics 7: 165-185.
- Shivakumar A, Rangappa D, Krishna H, Nagaraja P (2010) Development and kinetic validation of an assay for the quantitative determination of peroxidase: Application in the detection of activity in crude plant tissues. Enzyme Microb Technol 47: 243-248.
- Xia Y, Ye J, Tan K, Wang J, Yang G (2013) Colorimetric Visualization of Glucose at the Submicromole Level in Serum by a Homogenous Silver Nanoprism–Glucose Oxidase System. Analytical Chem 85: 6241-6247.
- Zhao H, Ju H (2006) Multilayer membranes for glucose biosensing via layer-by-layer assembly of multiwall carbon nanotubes and glucose oxidase. Analytical Biochem 350: 138-144.
- Dutta AK, Das S, Samanta S, Samanta PK, Adhikary B, et al. (2013) CuS nanoparticles as a mimic peroxidase for colorimetric estimation of human blood glucose level. Talanta 107: 361-367.
- Su L, Feng J, Zhou X, Ren C, Li H, et al. (2012) Colorimetric Detection of Urine Glucose Based ZnFe2O4 Magnetic Nanoparticles. Analytical Chem 84: 5753-5758.
- Wu P, He Y, Wang HF, Yan XP (2010) Conjugation of Glucose Oxidase onto Mn-Doped ZnS Quantum Dots for Phosphorescent Sensing of Glucose in Biological Fluids. Analytical Chem 82: 1427-1433.
- Liu Y, Yuan M, Qiao L, Guo R (2014) An efficient colorimetric biosensor for glucose based on peroxidase-like protein-Fe3O4 and glucose oxidase nanocomposites. Biosens Bioelectron 52: 391-396.
- Chen W, Chen J, Feng YB, Hong L, Chen QY, et al. (2012) Peroxidase-like activity of water-soluble cupric oxide nanoparticles and its analytical application for detection of hydrogen peroxide and glucose. Analyst 137: 1706-1712.
- Liu S, Tian J, Wang L, Luo Y, Sun X (2012) A general strategy for the production of photoluminescent carbon nitride dots from organic amines and their application as novel peroxidase-like catalysts for colorimetric detection of H2O2 and glucose. RSC Adv 2: 411-413.
- Zhang W, Ma D, Du J (2014) Prussian blue nanoparticles as peroxidase mimetics for sensitive colorimetric detection of hydrogen peroxide and glucose. Talanta 120: 362-367.
- Khalil OS (1999) Spectroscopic and Clinical Aspects of Noninvasive Glucose Measurements. Clinical Chem 45: 165-177.
- Purvinis G, Cameron BD, Altrogge DM (2011) Noninvasive Polarimetric-Based Glucose Monitoring: An in Vivo Study. J Diabetes Sci Technol 5: 380-387.
- Wróbel MS (2016) Non-invasive blood glucose monitoring with Raman spectroscopy: prospects for device miniaturization. IOP Conference Series: Materials Science and Engineering 104: 012036.
- Zuomin Z, Myllyla R (2001) Photoacoustic determination of glucose concentration in whole blood near infrared laser diode. Proceedings of SPIE 4256: 77-83.
- Zhang Y, Wu G, Wei H, Guo Z, Yang H, et al. (2014) Continuous noninvasive monitoring of changes in human skin optical properties during oral intake of different sugars with optical coherence tomography. Biomed Optics Express 5: 990-999.
- Pleitez MA, Hertzberg O, Bauer A, Seeger M, Lieblein T, et al. (2015) Photothermal deflectometry enhanced by total internal reflection enables non-invasive glucose monitoring in human epidermis. Analyst 140: 483-488.
- Peeling RW, Holmes KK, Mabey D (2006) Rapid tests for sexually transmitted infections (STIs): the way forward. Sexually Transmitted Infections 82: v1-v6.
- Clarke WL, Cox D, Gonder-Frederick LA, Carter W, Pohl SL (1987) Evaluating Clinical Accuracy of Systems for Self-Monitoring of Blood Glucose. Diabetes Care 10: 622.
- Akyilmaz E, Dinckaya E (2000) A mushroom (Agaricus bisporus) tissue homogenate based alcohol oxidase electrode for alcohol determination in serum. Talanta 53: 505-509.
- Chaubey A, Gerard M, Singhal R, Singh VS, Malhotra BD (2001) Immobilization of lactate dehydrogenase on electrochemically prepared polypyrrole–polyvinylsulphonate composite films for application to lactate biosensors. Electrochimica Acta 46: 723-729.
- Doretti L, Ferrara D, Lora S, Schiavon F, Veronese FM (2000) Acetylcholine biosensor involving entrapment of acetylcholinesterase and poly(ethylene glycol)-modified choline oxidase in a poly(vinyl alcohol) cryogel membrane. Enzyme Microb Technol 27: 279-285.
- Pilone MS, Pollegioni L (2002) D-amino Acid Oxidase as an Industrial Biocatalyst. Biocatal Biotransformation 20: 145-159.
- Toyama MH, Toyama D, Passero LFD, Laurenti MD, Corbett CE, et al. (2006) Isolation of a new l-amino acid oxidase from Crotalus durissus cascavella venom. Toxicon 47: 47-57.
- Çubukçu M, Timur S, Anik Ü (2007) Examination of performance of glassy carbon paste electrode modified with gold nanoparticle and xanthine oxidase for xanthine and hypoxanthine detection. Talanta 74: 434-439.
- Usman Ali SM, Alvi NH, Ibupoto Z, Nur O, Willander M, et al. (2011) Selective potentiometric determination of uric acid with uricase immobilized on ZnO nanowires. Sens Actuators B Chem 152: 241-247.
- Li G, Liao JM, Hu GQ, Ma NZ, Wu PJ (2005) Study of carbon nanotube modified biosensor for monitoring total cholesterol in blood. Biosens Bioelectron 20: 2140-2144.
- Nag S, Saha K, Choudhuri MA (2000) A rapid and sensitive assay method for measuring amine oxidase based on hydrogen peroxide–titanium complex formation. Plant Sci 157: 157-163.
- Batra B, Lata S, Rana JS, Pundir CS (2013) Construction of an amperometric bilirubin biosensor based on covalent immobilization of bilirubin oxidase onto zirconia coated silica nanoparticles/chitosan hybrid film. Biosens Bioelectron 44: 64-69.
- Rahman MA, Kwon NH, Won MS, Choe ES, Shim YB (2005) Functionalized onducting polymer as an enzyme-immobilizing substrate: An Amperometric glutamate microbiosensor for in vivo Measurements. Anal Chem 77: 4854-4860.
Open Access Journals
- Aquaculture & Veterinary Science
- Chemistry & Chemical Sciences
- Clinical Sciences
- Engineering
- General Science
- Genetics & Molecular Biology
- Health Care & Nursing
- Immunology & Microbiology
- Materials Science
- Mathematics & Physics
- Medical Sciences
- Neurology & Psychiatry
- Oncology & Cancer Science
- Pharmaceutical Sciences